 Want to receive Best Practice Bulletin directly to your inbox?
Sign up here.
Want to receive Best Practice Bulletin directly to your inbox?
Sign up here.
Published: 29 July, 2022
Contents
New article - H. pylori: who to test and how to treat
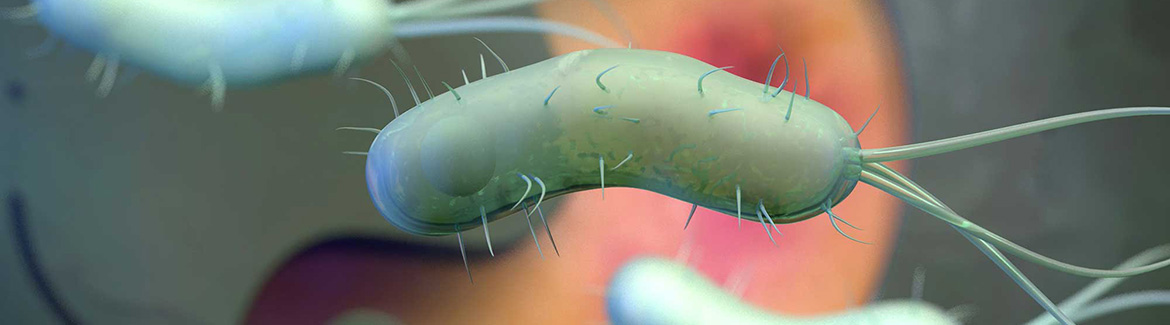
Helicobacter pylori is a gram-negative bacterium associated with long-term gastric inflammation. The prevalence in New Zealand is lower than in many other parts of the world, but this is still a potential factor in people presenting with dyspepsia-like symptoms: particularly those of non-European ethnicity. In this article we discuss the risk-based approach to investigation for H. pylori, along with the recommended test and treatment regimen.
Read the full article here.
Coming up next - Coeliac disease: not just a gut feeling
COVID-19 health notifications
There are increasing reports in the media that we have now reached the peak of case numbers in the latest COVID-19 wave. This, however, is not necessarily reflected in the number of people seeking healthcare services. It is also highly likely that many people are not testing for, or reporting, infections. More indicative measures of the true burden of infection include hospitalisations and wastewater test results.
Wastewater data:
ESR has developed an interactive dashboard that displays national and regional surveillance data on COVID in wastewater samples. This allows users to track COVID risk in specific areas of the country. In most regions there is currently a marked discrepancy between reported cases and wastewater results. Click here.
Ministry of Health notifications:
Health professionals can subscribe to an email newsletter that covers key health messages about the COVID-19 response. A list of other Manatū Hauora (Ministry of Health) newsletters that health professionals can subscribe to is available here.
Āwhina is a Ministry of Health app that provides COVID-19 information for people working in the health sector, e.g. case numbers, updated guidance for the aged care sector; it is available for free download here.
N.B. We note that recent clinical changes, e.g. changes to eligibility criteria for antivirals and vaccine boosters, were not announced first via the methods of communication above.
Guidance for recovery from acute COVID-19
As the cumulative number of people who have had COVID-19 infection continues to increase in New Zealand, healthcare professionals will find themselves more frequently providing care to patients who have ongoing symptoms. The Ministry of Health has recently published a “living document” produced via a collaboration of allied health professionals, that “highlights the complexity and potential long-term needs” of people recovering from the acute phase of COVID-19. While this document does not directly cover the role of general practice, it gives insight into the types of issues that allied health professionals, e.g. physiotherapists, psychologists, social workers, can support these patients with in their rehabilitation. A separate document is currently being developed for long COVID rehabilitation.
Read more
Many people recover from the acute phase of COVID-19 without any notable health problems. However, for a small number of patients more serious health impacts can be experienced, including breathing disorders, post-viral fatigue syndrome, renal and hepatic injury, cognitive impairment, impaired swallowing and malnutrition. Social impacts may include fear of accessing healthcare (or difficulty in doing so), increased anxiety and loss of income.
This living guidance document sets out a framework for addressing these specific health and social impacts in people experiencing ongoing issues in recovery from COVID-19.
Definitions of clinical phases of COVID-19:
| Acute COVID-19 |
Signs and symptoms of COVID-19 for up to four weeks |
| Ongoing symptomatic COVID-19 |
Signs and symptoms of COVID-19 that persist after four weeks and up to twelve weeks |
| Post-COVID-19 syndrome (“long COVID”) |
Signs and symptoms that develop during or after an infection of COVID-19 that continue for more than twelve weeks and are not explained by an alternative diagnosis |
Ocular decongestant drops should not be used in children
Medsafe is currently consulting on a proposal to add warning statements to ocular decongestant eye drops to prevent inappropriate use in children. This serves as a general reminder to pharmacists and other health professionals that eye drops containing naphazoline (e.g. Clear Eyes) should not be used in children aged under 12 years: some packaging does not state this precaution.
Warning over adulterated herbal product marketed for inflammation
Medsafe has issued a warning about a herbal product being marketed and sold in New Zealand via social media and other channels, that has been found to contain at least three prescription medicines. Nhan Sam Tuyet Lien Truy Phong Hoan is promoted using therapeutic claims that it is useful for the treatment of gout, arthritis, lower back pain and other inflammation. It contains furosemide, chlorpheniramine and dexamethasone (which is not stated in product information). Any patients taking this medicine should be advised to stop use immediately. N.B. the product and packaging is pictured within the Medsafe statement.
First annual report on assisted dying
The Assisted Dying – Ngā Ratonga Mate Whakaahuru service came into effect on the 7 November, 2021, as a result of the End of Life Choice Act (2019). The first annual report on this service, covering the period 7 November, 2021 – 31 March, 2022, has now been published. The report covers how the service was implemented, the groups, organisations and workforce involved, the people who have applied to access the service, and the feedback to date.
Key facts
The service
- The framework for delivery of the service is based on the Standard of Care, available here, and Clinical Guideline (only available to practitioners directly involved in the service)
- Te Apārangi: Māori Partnership Alliance provides governance and strategic oversight to the service
- There are two hospital pharmacies in New Zealand contracted to provide medicines for assisted dying
- Funding is available for practitioners to travel to provide services so people can receive care regardless of their location; this also allows people choice of practitioner
- As of 31 March, 2022 there were 130 practitioners registered with the service (91 as attending medical practitioners)
- All reports on completed assisted deaths are reviewed by the End of Life Review Committee, which is comprised of a medical ethicist, a doctor specialising in end-of-life care and one other health practitioner; one member is Māori
Applicants
- Between 7 November, 2021 and 31 March, 2022, there were 206 applications for assisted dying
- 66 people had an assisted death
- 59 people were still in the process of assessment (25) or preparation (34) for assisted death
- 81 people did not continue the process due to ineligibility (40), withdrawal (11) or death due to their condition (30)
- Ethnicity: Of the 206 applicants, 79% were NZ European, 6% were Māori, 2% were Asian and 13% were other ethnicities (none were Pacific)
- Age and sex: Of the 206 applicants, 55% were female, 74% were aged ≥ 65 years
- Health status: Of the 206 applicants, 65% had a cancer diagnosis, 10% had a neurological condition, 80% were receiving palliative care at the time of application
- Location of death: 73% of assisted deaths occurred at the persons home or other private residence and 17% in aged care facilities
Paper of the Week: Fever therapy
Starve a fever, feed a cold…hang on, is that right? And does it just mean no chicken soup or is it no medicines either? Although we can safely relegate this saying into the annals of “Old Wives Tales”, there has been much debate from actual clinical professionals over the role of antipyretics in reducing fever – is this an essential treatment, or does this practice impair the body’s ability to fight off the infection?
This question has been answered in the latest edition of BMJ with a meta-analysis and systematic review of 42 clinical trials. We will go through the results below, but for those of you who love a spoiler…keep on feeding that fever (with antipyretics), or not, it doesn’t seem to make much difference either way.
Read more
- The physiological and behavioural responses that occur in response to fever cause metabolic stress and increased oxygen demand, in severe cases leading to seizure and organ failure, therefore the clinical rationale is that reducing fever will improve patient outcomes
- However, fever also induces cellular and immunological changes that are part of the inflammatory response against infection, and some studies have found that antipyretics increase the duration of some infections
- The authors conducted a systematic review with meta-analyses and trial sequential analyses of 42 randomised clinical trials including 5140 participants
- The trials assessed antipyretic medicines (mainly paracetamol and ibuprofen), physical cooling and combination treatment v.s. no fever treatment or placebo, in patients with fever who were critically ill, non-critically ill, with and without infectious illness
- There was no evidence that fever therapy reduced the risk of death (risk ratio 1.04 [0.9 – 1.19]) or the risk of serious adverse events (risk ratio 1.02 [0.89 – 1.17]); conversely there is no evidence that fever therapy increases these risks
- The authors concluded that: Fever therapy does not seem to affect the risk of death and serious adverse events.
- There was insufficient evidence to determine whether fever therapy influences quality of life or non-serious adverse events – we can’t even say for certain that at least it will make the patient feel better
Holgersson J, Ceric A, Sethi N et al. Fever therapy in febrile adults: systematic review with meta-analyses and trial sequential analyses. BMJ 2022;378:e069620. doi: https://doi.org/10.1136/bmj-2021-069620
 This Bulletin is supported by the South Link Education Trust
This Bulletin is supported by the South Link Education Trust
If you have any information you would like us to add to our next bulletin, please email:
editor@bpac.org.nz
© This resource is the subject of copyright which is owned by bpacnz. You may access it, but you may not reproduce it or any part of it except in the limited situations described in the terms of use on our website.
