How did the problem with oxycodone evolve?
When oxycodone was first introduced into New Zealand in the early 2000s, it was regarded by many as a “new and improved”
strong analgesic, with fewer adverse effects and perhaps none of the stigma associated with morphine. As a result, prescribing
of oxycodone increased significantly over the next few years, reaching its peak in 2011/12. The number of prescriptions
for morphine remained relatively stable over this same time period, suggesting that a new patient population being treated
with oxycodone had been created.
Paralleling this surge in oxycodone use, reports of misuse and addiction emerged in New Zealand, following the trend
observed in other countries with a longer history of oxycodone use. It has now become apparent that there is little or
no advantage of oxycodone over morphine in terms of managing pain. Oxycodone is associated with the same adverse effects
as morphine, and appears to be even more addictive than morphine. Therefore, there is no reason to continue to prescribe
oxycodone instead of morphine (unless intolerable adverse effects have occurred with morphine), or to prescribe it when
a less potent analgesic would be more appropriate.
The Wellington psychiatrist and addiction specialist Dr Jeremy McMinn commented
in his interview in BPJ 61 (Jun, 2014), that in terms of the misuse of oxycodone in New Zealand, we are “looking
at a disaster in the making”. Clinicians are urged to assess whether oxycodone is appropriate when initiating or continuing
a prescription and, if necessary, make changes to their prescribing behaviour. How is it best to manage the problem with
oxycodone? According to Dr McMinn: “Don’t get there in the first place”.
The international experience
Oxycodone was first synthesised in Germany in 1916 and became available for use in the United States in 1939. For many
years it was used overseas as a component of combination short-acting analgesics, including paracetamol and NSAIDs. The
controlled-release oxycodone-only formulation, OxyContin, was approved by the Food and Drug Administration (FDA) in the
United States in 1996. In New Zealand, oxycodone was approved by Medsafe in 2001 and the oral forms (controlled and immediate-release)
were subsidised on the New Zealand Pharmaceutical Schedule from 2005.
Since its release, the use of oxycodone has increased dramatically and many countries are now dealing with misuse and
addiction issues. For example, in Ontario, Canada, the number of prescriptions for oxycodone increased by 850% between
1991 and 2007.1 After controlled-release oxycodone was added to the Ontario state drug formulary there was
a five-fold increase in oxycodone-related mortality, along with a 41% increase in overall opioid-related mortality.1 Similar
increases in the prescribing rates for oxycodone have also been observed in the USA. The national estimates for drug-related
emergency department visits for oxycodone-containing medicines increased from 27.6 per 100 000 people in 2004 to 88.5
visits per 100 000 in 2009.2 In Australia, the oxycodone supply increased 22-fold between 1997 and 2012, and
oxycodone became the seventh most commonly prescribed medicine in general practice.3 By 2007, a national sample
of injecting drug users found that 51% had reported using oxycodone.4
The situation in New Zealand
Between 2008 and 2013, the number of dispensed prescriptions for strong opioids in New Zealand has increased significantly
(Figure 1). Much of this increase is attributed to a growing number of dispensed prescriptions for
oxycodone. The most recent dispensing data from 2013 suggests that the number of prescriptions for oxycodone may be reaching
a plateau, but the fact remains that oxycodone is a second-line option for moderate to severe pain, and should be dispensed
considerably less than morphine.
The type of prescribers initiating oxycodone remained similar in 2013, compared with when first reported in 2011. Approximately
30% of prescriptions for oxycodone are written by General Practitioners and the remaining 70% are from other clinicians,
e.g. those working in secondary care.5 In 2013, the proportion of prescriptions initiated in secondary care
and continued in general practice was 17%, the same figure as in 2011 (Figure 2).5
Given the current best practice recommendations that oxycodone generally be reserved for second-line treatment after
morphine, it is concerning that approximately 80% of patients prescribed oxycodone for the first time in 2013
did not have a previous prescription for morphine in the preceding 12 months.5 This suggests that
the majority of first-time prescribing of oxycodone is occurring before a trial of morphine, or alternatively, patients
are being treated in hospital with parenteral morphine or pethidine and discharged with oral oxycodone.
 For further information see: "Oxycodone
use still increasing", BPJ 36 (Jun, 2011). "Update on Oxycodone what can primary
care do about the problem", BPJ 44 (May 2012).
For further information see: "Oxycodone
use still increasing", BPJ 36 (Jun, 2011). "Update on Oxycodone what can primary
care do about the problem", BPJ 44 (May 2012).
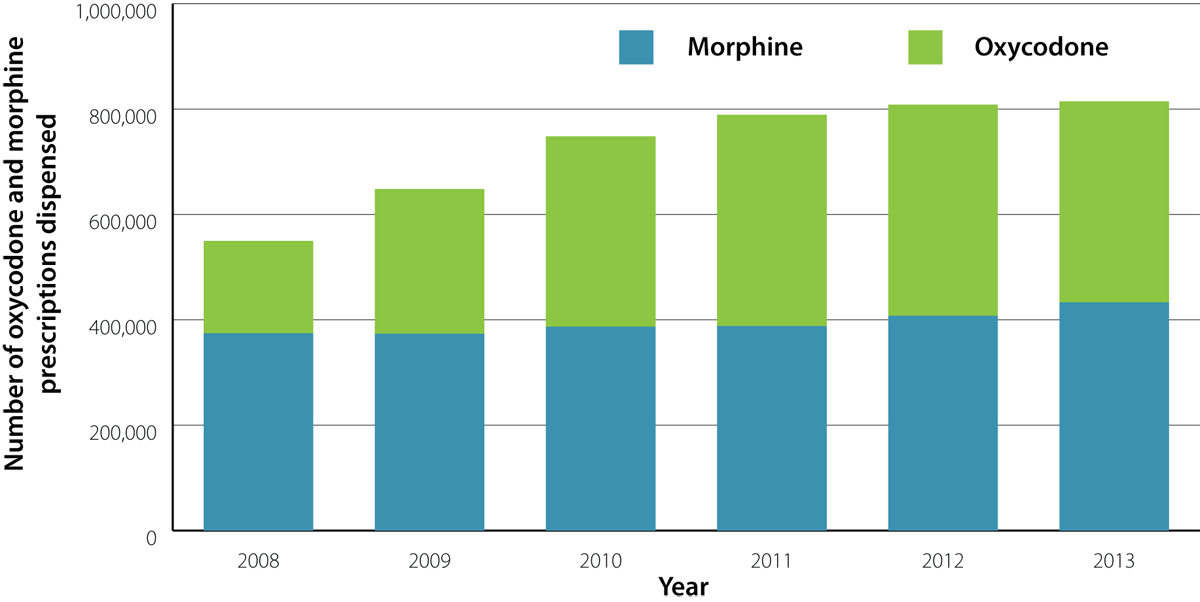
Figure 1: Number of oxycodone and morphine prescriptions dispensed (excluding injected preparations).5
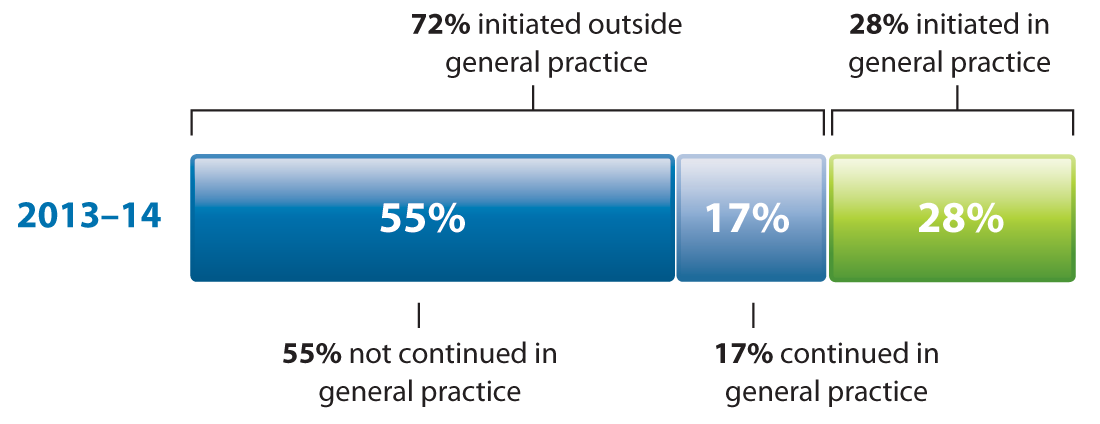
Figure 2: Source of oxycodone prescriptions for patients initiated in 2013/14 (Apr 2013 – Mar 2014)
The evidence about oxycodone
The strong marketing of oxycodone (See: “The oxycodone marketing campaign), along with its rapid
rise in popularity, means that many aspects of its pharmacology and general use may be misunderstood. There are few head-to-head
trials comparing oxycodone with morphine or other opioids, yet several claims have been made about its alleged superiority,
many of which are not entirely accurate. There is no debating that oxycodone is an effective analgesic, however, there
is no compelling clinical reason to choose it over morphine, and the associated risks and problems with its use, clearly
place oxycodone as a second-line option.
Potency: oxycodone is a strong opioid
Despite its name, oxycodone is not a “natural version of codeine” or a “gentle analgesic” – it is approximately twice
as potent as morphine, i.e. 10 mg of oxycodone is equivalent to 15–20 mg of morphine.6, 7 A clinical trial
reported that oral controlled-release oxycodone was twice as potent as oral controlled-release morphine in patients who
received single doses for post-operative pain following hysterectomy.8 For total and peak analgesic effects,
the doses of 20 mg and 40 mg oxycodone were comparable to morphine doses of 45 mg and 90 mg, respectively.8
Oxycodone is approximately 7.5 – 20 times more potent than codeine, i.e. 10 mg of oxycodone is equivalent to 75-200
mg of codeine.6
The stigma associated with morphine is a reason that some patients are reluctant to use it, however, the same patients
are comfortable using oxycodone. When discussing appropriate analgesic treatments with patients, clinicians need to ensure
that patients understand that oxycodone is used for the same purpose as morphine and is actually more potent.
Addictive potential: oxycodone rates higher than morphine
All opioid analgesics (including weak opioids) are potentially addictive, but the marketing campaign for oxycodone promoted
the belief that it had a lower addictive potential than other strong opioids. However, the literature suggests that oxycodone
actually has a higher addictive potential than morphine.
A systematic review of nine randomised trials compared the likeability and likelihood of misuse of oral oxycodone, morphine
and other selected opioids in recreational drug users and people with a history of opioid misuse. It was found that oxycodone
was more favoured and more likely to be misused than either morphine or hydrocodone (not available in New Zealand).9 Oxycodone
demonstrated high subjective attractiveness ratings with a few negative ratings across the majority of studies included
in the review. Oxycodone was also associated with consistently higher “take again” ratings than morphine.9
The addictive potential of strong opioids needs to be discussed with and understood by patients before they are prescribed.
A psychological assessment of the likelihood of addiction forms part of the risk-benefit analysis for the decision to
prescribe an opioid.
Renal impairment: use oxycodone with caution
Many clinicians have prescribed oxycodone in preference to morphine due to the belief that oxycodone is safer in patients
with renal impairment. However, oxycodone should be used with caution in patients with renal failure and prolonged use
avoided in patients with an eGFR < 10 mL/min/1.73 m2 (due to the risk of accumulation of metabolites).10 Case
reports of oxycodone toxicity in patients with renal impairment have been reported, along with increased sedation.11
In many cases, morphine can still be safely used in patients with renal impairment, if it is dosed carefully; use the
lowest effective dose and consider the cumulative effect. Patients will have an individual response to morphine in terms
of its effect on their renal function.
Fentanyl is regarded as the safest strong opioid for patients with renal impairment (although does have other adverse
effects).10 Methadone is also an appropriate option for patients with renal impairment, but can be complex
to dose and should only be prescribed if the clinician is familiar with its use.10 Codeine, pethidine and
tramadol should be avoided in people with renal impairment.
The two main metabolites of oxycodone are oxymorphone (a very potent analgesic) and noroxycodone (a weak analgesic),
which are both renally excreted.6 It is reported that up to 19% of oxycodone is eliminated unchanged in the
urine.12 There is limited data on renal clearance of oxycodone. A small study in people with mild-to-moderate
renal dysfunction showed that the peak plasma oxycodone and noroxycodone concentrations were approximately 50% and 20%
higher, respectively, than in people without renal failure.13 The AUC (area under the curve – a measure of
total exposure to a drug) values for oxycodone, noroxycodone and oxymorphone were approximately 60%, 50% and 40% higher
in people with renal dysfunction than in people with normal renal function, respectively.13
 For further information see: “Fentanyl patches to be available without
Special Authority in 2011”, BPJ 33 (Dec, 2010). “Methadone – safe and effective use for chronic pain”
BPJ 18 (Dec, 2008).
For further information see: “Fentanyl patches to be available without
Special Authority in 2011”, BPJ 33 (Dec, 2010). “Methadone – safe and effective use for chronic pain”
BPJ 18 (Dec, 2008).
Adverse effect profile: similar overall to morphine
Overall, oxycodone and morphine have similar adverse event profiles that are consistent with other opioid analgesics.
The most common adverse events reported with use of oxycodone (with approximate rates) are constipation (25–30%), nausea
(25%), drowsiness (15%), vomiting (10–15%) and pruritis (10–15%).6 It has been reported that constipation
occurs more frequently in people taking oxycodone compared to those taking morphine.6 The incidence of the
other adverse effects are generally similar, however, a few studies have reported that the rates of nausea and vomiting,
hallucinations and pruritis may be lower in people taking oxycodone compared to those taking morphine.6
Oxycodone, unlike morphine, is mainly metabolised by the CYP3A and CYP2D6 enzymes in the liver.7 The involvement
of CYP3A in the metabolism of oxycodone makes it more prone to interactions with drugs that inhibit or induce this enzyme.
7 Inhibitors
of CYP3A, e.g. ritonavir, clarithromycin, itraconazole, miconazole and grapefruit juice potentiate the effect of oxycodone,
resulting in an increased risk of adverse effects.7 Inducers of CYP3A, e.g. St John’s wort and rifampicin,
reduce exposure to oxycodone.7 This may result in people taking higher doses of oxycodone, which becomes problematic
if the enzyme-inducing medicine is stopped.
CYP2D6 is a highly polymorphic enzyme; gene mutations and deletions cause the enzyme to be non-functional or over-expressed.
This results in people having phenotypes for poor, intermediate, extensive or ultra-rapid metabolisers of drugs which
are dependent on this enzyme. Most evidence has found that the CYP2D6 genotype does not have a significant influence on
the analgesic effect of oxycodone or risk of adverse effects, but this is an ongoing area of research.7
Oxycodone prescribing in secondary care
Dispensing data shows that in New Zealand, the majority of prescriptions for oxycodone are not being written by General
Practitioners.5 This suggests that a considerable proportion of oxycodone is being prescribed to patients
on discharge from hospital, e.g. for post-surgical pain management. Some General Practitioners feel compelled to continue
this prescribing, therefore adding to the problem. Studies have suggested that hospital and specialist prescribing is
an important influence on General Practitioner’s prescribing behaviours.18, 19
The key messages for secondary care are:
- Avoid prescribing oxycodone instead of morphine in a hospital setting, unless the patient cannot tolerate morphine
- Consider whether it is appropriate to be sending a patient home with a strong opioid
- Do not give patients the expectation that a General Practitioner will continue a prescription for a strong opioid
once they are discharged. Emphasise that all opioids have the potential to be addictive and in most circumstances, they
are for short-term use only.
In response to this issue, South Australia’s Health Department (SA Health) has developed guidelines for prescribing
opioids on hospital discharge.20 Immediate-release opioids may be appropriate on discharge if they have been
newly commenced for acute or breakthrough pain in hospital, and are still required. Slow-release opioids should only
be prescribed on discharge if the patient was already taking long-term opioids prior to their hospital admission, and
their dose requirements have changed. Patients commenced on long-term opioids in hospital for chronic pain, e.g. cancer
pain, should receive appropriate follow-up on discharge from their hospital specialist or General Practitioner.20
The SA Health guidelines suggest that the following points are considered when determining whether to prescribe an
opioid on discharge:20
- Review the patients opioid requirements over the 24 hours prior to discharge
- Patients with acute non-malignant pain whose opioid requirements have not reduced during their admission may not
yet be ready for discharge
- The discharge prescription dose should not exceed the patient’s dose administered in hospital
- The dose should be calculated based on the preceding 24 hours in hospital, not the patient’s initial analgesic requirements
- Prescribe a quantity appropriate to the patient’s anticipated requirements (usually no more than enough for three
days or 20 pills)21
An additional point to consider (not included in the guidelines) is whether a strong opioid is still required or whether
it may be more appropriate to prescribe a weaker opioid on discharge, such as codeine.
The patient should be given clear instructions on the use of analgesics they are prescribed, the adverse effects they
may expect and a pain management plan. It is recommended that if a patient is discharged with a prescription for an opioid,
this is communicated to the patient’s General Practitioner, including information on opioid dose frequency, suggested
duration of treatment and plan for dose reduction.20
The patient should be reviewed by their General Practitioner within three to five days.21 The aim should
be to step down to other forms of analgesia, such as a weaker opioid (e.g. codeine), an NSAID or paracetamol when possible.21 The
decision to continue strong opioids should only be made after an assessment of the cause of pain and why it is not resolving
and a discussion about the risks and benefits to the patient of continuing treatment.21
Best practice points for the use of opioids for acute pain:21
- Maximise appropriate non-opioid treatments
- Use a shared decision making approach and ensure the patient is educated about the risks and benefits of opioid
treatment
- Avoid prescribing more than three days’ supply or more than 20 pills of low-dose, short-acting opioids unless circumstances
clearly warrant additional opioid treatment
- Prescribe opioids with caution in elderly patients: take into account renal function and consider prescribing lower
doses
- Follow up with the patient within three to five days to assess the response to treatment and any adverse events
- Make sure the patient is aware that opioids can affect their work duties and driving
- Ensure the patient is aware about storing opioids in a secure place away from children, and safe disposal
What lessons can be learnt?
The New Zealand statistics show that although the growth in oxycodone prescriptions may have slowed in recent years,
prescribing rates are still very high. The data from Canada, the USA and Australia regarding illicit use, hospitalisations
and deaths as a result of oxycodone should be of great concern to New Zealand as these countries have a longer experience
with oxycodone use. The overriding message is that continued high prescribing rates will eventually result in more illicit
use of oxycodone, more people addicted to oxycodone, and associated downstream effects, which New Zealand is already starting
to see (this will be examined in further detail in the next article in this series).
Key points for reducing the use of oxycodone:
- Morphine is the first-line treatment when a strong opioid is indicated for moderate to severe pain; this applies
in any setting
- Oxycodone is not an appropriate analgesic for mild to moderate pain
- If patients are discharged from hospital with a strong opioid, the prescription should cover a short time period
only and the patient should have a treatment plan for tapering use of analgesics
- Primary care clinicians do not need to repeat a prescription for patients discharged from hospital on a strong opioid
- The decision to prescribe oxycodone, or any strong opioid, should take into account the predicted net benefits from
treatment, weighed up with the risks of adverse effects, misuse and addiction
Appendix B – “Scripting Support for Saying No to a Patient and an Opioid Prescription” may be particularly
useful for primary care clinicians.
Acknowledgement
Thank you to Dr Kieran Davis, Clinical Director, Auckland Regional Pain Service and Dr Peter
Jones, Rheumatologist and Chief Advisor, Sector Capability and Implementation, Ministry of Health for expert
review of this article