Published: 20th March, 2026
Reporting suspected adverse reactions enables Medsafe to quickly identify and respond to emerging medicine safety issues. If a safety concern is confirmed, Medsafe can take a range of actions, such as updating medicine data sheets, issuing safety alerts, requiring package warnings or even changing the classification of a medicine.
New Zealand has historically had a high rate of adverse reactions reporting, and healthcare professionals have an important role in helping Medsafe identify medicines safety issues. Primary care clinicians are in an excellent position to identify and report adverse reactions because of the trusted relationships they have with patients and the continued involvement in their health.
What happens to my report?
All adverse drug reaction reports are collected and processed into a database by Medsafe. Non-routine reports (e.g. a new or serious potential adverse effect) are medically assessed by physicians at CARM to help prioritise those reactions which look most likely to have been caused by the medicine (causality assessment).
The reports in the database are used for detection of safety issues through review of individual cases highlighted by the causality assessment and through a statistical disproportionality analysis. Information from relevant cases is used to inform Medsafe’s evaluations and actions on safety issues.
An example of medicines safety monitoring in action is the adverse effect of Fournier’s gangrene identified with empagliflozin. A series of local reports in 2022 showed a high proportion of cases in Māori and Pacific peoples, highlighting a need for further action. This prompted Medsafe and CARM to promote awareness of Fournier’s gangrene as an adverse effect of empagliflozin among healthcare professionals. Medsafe also created an information leaflet for patients in English, Te Reo and Samoan.
Reporting a reaction also allows CARM to place a danger alert on the National Medical Warning System for the patient; currently this information can be accessed by hospital clinicians but access is not yet widely available in primary care. These alerts inform healthcare professionals of known risk factors, such as allergies or previous serious adverse reactions, that may be critical when making clinical decisions about a patient (e.g. when a patient is transferred to hospital care).
The adverse reaction tool was designed by BPAC Clinical Solutions to make the reporting of adverse reactions easier and less time consuming. It uses an online reporting form pre-populated with patient details from the Practice Management System (PMS). This reduces manual entry of information.
The ability to extract data from PMS software makes it easier to include more details such as laboratory test results and other investigations. This improves Medsafe and CARM’s ability to review the reports and detect potential safety issues. A copy of the report is added to the patient’s electronic record.
How do I use the reporting tool?
To access the reporting tool, look for ‘Adverse Drug Reaction Reporting’ on the modules list on your BPAC Dashboard.
Once opened, the tool automatically pre-populates the patient’s medical history, recent prescribed medicines and gives the option of including laboratory test results.
As vaccines make up approximately one-third of the adverse reaction reports received every year, the tool has been designed with a specific vaccine tab. If the suspected medicine is a vaccine, the tool pre-populates the batch number, the date of administration and how the vaccine was given.
Once a description of the reaction and other pertinent information is entered, the report is electronically sent to Medsafe. The details of the patient and reporter are encrypted in the electronic reporting tool and the information provided in the report is only viewed and used by Medsafe and CARM. De-identified information is sent to the World Health Organization (WHO) as part of Medsafe’s international obligations.
Including your email address in the report will speed up correspondence if Medsafe or CARM have any follow-up questions.
By the numbers: use of the electronic ADR tool
Over 18,000 adverse drug reaction reports have been made using the BPAC Clinical Solutions ADR online reporting tool since its launch by the Minister of Health in 2009 (Figure 1). Over the first decade of use there were on average just under 1,200 reports made per year. Reports began to decline slightly before peaking again during the COVID-19 pandemic – likely due to vaccine-related reports. Since 2023, numbers have reduced and last year only 341 reports were made, suggesting that awareness of the tool may be waning.
Data on overall adverse drug reaction reporting in New Zealand* is currently available up to 2024; there were 4,585 adverse drug reaction reports in 2024 of which 1,569 related to a vaccine. There were 510 reports using the electronic ADR tool in 2024, representing 11% of ADR reports. The peak use of the ADR tool was in 2021 with 1,939 reports, which accounted for 48% of the total report in New Zealand that year (4,006)†.
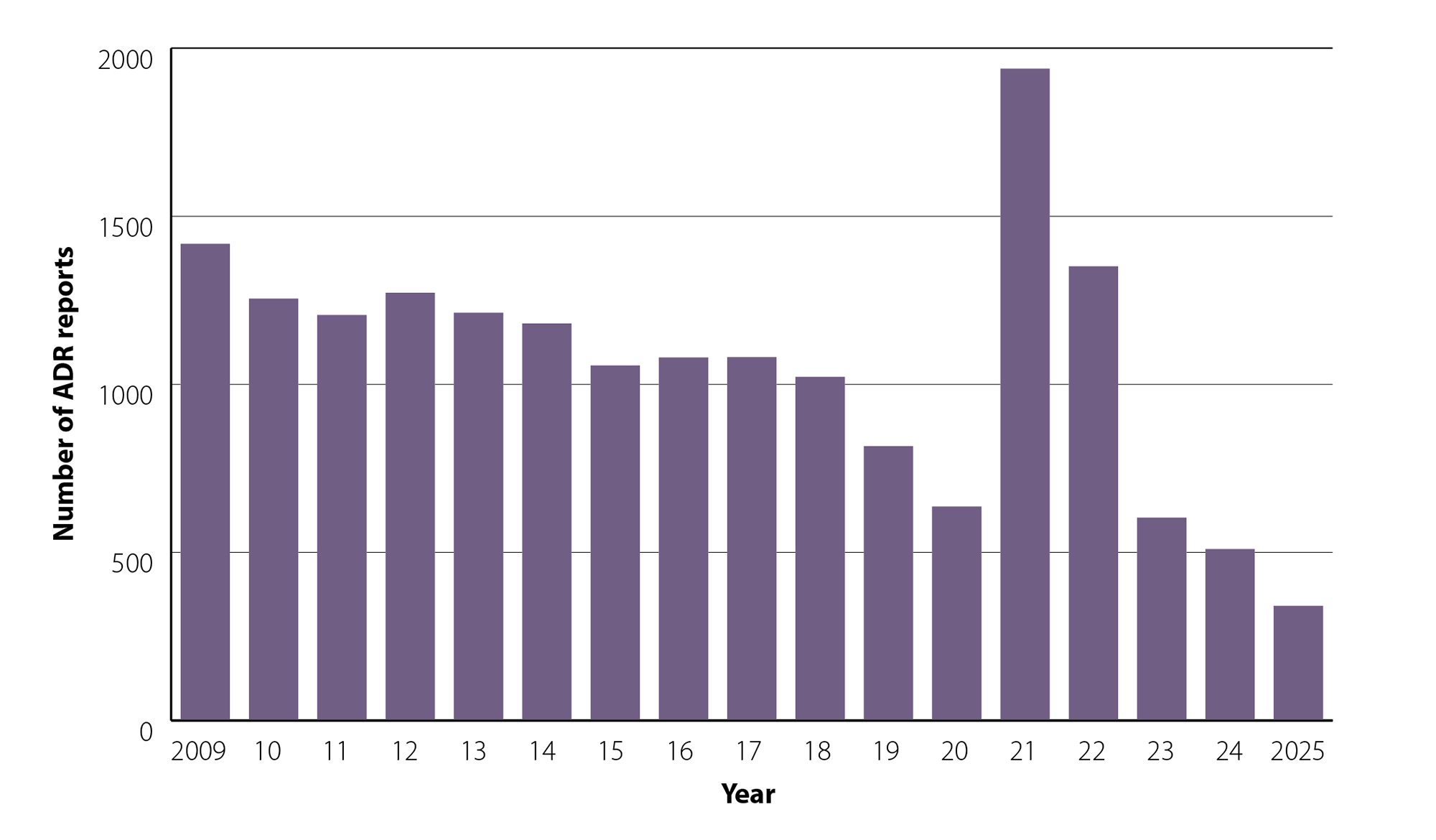
Figure 1: Number of ADR reports per year using the BPAC Clinical Solutions ADR reporting tool.
* ADR data available from: https://www.medsafe.govt.nz/safety/reports-and-promotion/ADR-reporting-statistics.asp. Prior to 2023, influenza vaccination ADRs were reported separately, and from 2023 onwards, COVID-19 vaccines were included in the overall number of reports.
† In 2021 there were 3,581 ADR reports and 425 reports following influenza vaccination
Other ways of reporting
If you do not have access to the BPAC Clinical Solutions ADR reporting tool, you can still make an adverse reaction report, e.g. using online New Zealand Adverse Reactions Reporting Form. However, unlike reporting using the ADR electronic tool, patient data will not be pre-populated, therefore this process will take more time.
The report must include:
- A patient identifier
- The suspect medicine
- Details of the adverse reaction
- Reporter details
Additional information isn’t compulsory but is important to support the evaluation of safety concerns. For serious adverse reactions, ideally, include:
- Medicine start and stop dates
- Date of onset and clinical course of the adverse reaction
- Concomitant medicines, co-morbidities and medical history
- Medicine dosages
- Any action taken and what happened
Where can I find other adverse reaction reports?
Reports that highlight significant safety issues with medicines and how they are used are described in the “Gathering knowledge from adverse reaction reports” article in each quarterly issue of Medsafe’s Prescriber Update.
You can also look for reports using Medsafe’s Suspected Medicine Adverse Reaction Search (SMARS). Adverse reaction reporting statistics are also published on the Medsafe website each year.
Need help?
If your practice does not have access to the BPAC dashboard or the Adverse Drug Reaction reporting tool, enquire about signing up for free here.
If you have any questions about using the tool, email: itservicedesk@bpacsolutions.com
If you would like to know more about adverse reaction reporting, email the Medsafe Pharmacovigilance Team: CARMreport@health.govt.nz





