* Amfetamine is the International Non-Propriety Name (INN) and is the term/spelling used in New Zealand Universal List of Medicines (NZULM) and the New Zealand Formulary (NZF)
Confirmed ADHD diagnosis is currently more prevalent among males during childhood, however, this sex disparity is reducing as more adults seek diagnosis.1, 3 Inattentive-type ADHD predominates in females and is often not recognised until later in life (or is misdiagnosed).1, 4 It has also been hypothesised that puberty-related changes in sex hormones affect emotional regulation and may worsen ADHD symptoms in females leading to a delayed diagnosis estimated to be up to four years later than males.4 A combination of these factors likely contributes to the differences observed in dispensing of psychostimulant medicines for ADHD between males and females in New Zealand (Figure 1). Barriers to medical and psychological care and assessment may also lead to variations in prevalence across ethnicities (see: “A snapshot of psychostimulant medicine dispensing in New Zealand”).
The concept of “adult- or late-onset ADHD” as a separate clinical condition is controversial. The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR) diagnostic criteria for ADHD requires symptom onset before age 12 years.5 However, symptoms may not be identified in some people during childhood due to factors such as lack of awareness among parents and teachers, lack of access to appropriate services for assessment, the symptoms initially being subclinical (but then worsening over time) or attributed to another cause.3 It is also possible that parents and teachers established appropriate scaffolding (i.e. structured routines, organisational tools and environmental supports) around the person with ADHD enabling them to successfully compensate for or mask their symptoms.1, 6 Symptom masking must be considered in the context of the DSM-5-TR criteria for diagnosing ADHD which require that symptoms are pervasive, occurring in two or more settings, including social, familial, educational and/or occupational settings.5
Previously, neither general practitioners nor nurse practitioners working in primary care were permitted to initiate or continue prescribing methylphenidate, lisdexamfetamine or dexamfetamine in adults or children, without written approval from a psychiatrist or paediatrician.17 Primary care clinicians also required separate written approval when switching between psychostimulant medicines. Since 1st December, 2024, there has no longer been a requirement to renew Special Authority approvals for psychostimulant medicines; once issued they remain valid indefinitely.18
The introduced regulatory and funding changes mean that ADHD diagnosis and management is likely to extend more into the primary care setting over time. However, presently many primary care clinicians and practices will not have the resources or experience to offer this service for their patients. There is no requirement for primary care clinicians to upskill in the management of patients with ADHD, but facilitating access to treatment will address some of the barriers that are currently experienced by patients, e.g. wait times and availability of publicly funded psychiatry services and the cost of these services privately (see: “Addressing inequities in accessing psychostimulant medicines for ADHD”).19
As Dr Michael Buckley, a general practitioner with a specific interest in ADHD puts it: “There is already high demand for ADHD assessment in both adults and children for a number of reasons (increased public awareness, barriers to timely assessment) and there will be mismatch between what patients expect the regulatory changes mean and what primary care clinicians can currently deliver. The only change that is happening is that certain primary care clinicians with the appropriate extra training and expertise can initiate stimulant treatment for patients with a confirmed diagnosis of ADHD. The need for, and elements of a comprehensive assessment to establish the diagnosis of ADHD do not change.”
ADHD diagnosis is complex; symptoms overlap with other mental health conditions, and co-morbidities can influence the level of impairment people experience.3, 20 A comprehensive assessment for ADHD is required, involving collection of clinical information about the patient’s social, family, educational and/or occupational history, current circumstances and presenting difficulties, results of psychometric tools, and collateral information from multiple respondents, e.g. teachers, employers;1 this process requires more than a conventional 15 minute primary care consultation. The cost and time implications for clinicians’ professional development to upskill in the diagnosis of ADHD and the establishment of a fee-for-service model, have not been addressed at a national level. Therefore, these challenges will need to be navigated by individual general practice clinics, likely resulting in a variation of services and patient fees between regions.
N.B. Diagnosis of ADHD in primary care is outside the scope of this article. For further information on this topic, see: “Clinician resources”.
The pathophysiology of ADHD is not completely understood but likely involves disruption of dopamine and noradrenaline pathways in the prefrontal cortex, a region of the brain responsible for executive functioning processes, including attention and working memory.1, 21 This dysregulation results in reduced signal strength, contributing to hyperactive and inattentive symptoms (Figure 2).21 Psychostimulant medicines, e.g. methylphenidate and amfetamines, improve cognitive, emotional/affective and behavioural symptoms by increasing neurotransmitter activity in the prefrontal cortex.21 Methylphenidate and amfetamines primarily inhibit dopamine and noradrenaline transporters.21 Amfetamines also inhibit monoamine oxidase enzymes, preventing neurotransmitter breakdown, and regulate vesicular monoamine transporters (VMAT-2), modifying neurotransmitter reuptake and release.21 These actions increase extracellular dopamine and noradrenaline concentrations at the synaptic cleft, which is thought to improve hyperactive and inattentive symptoms by intensifying signal strength.21
The pharmacokinetics of the different formulations of funded psychostimulant medicines available in New Zealand are summarised in Table 2.
Methylphenidate hydrochloride is highly soluble and rapidly absorbed in the small intestine.22 Hepatic carboxylesterase enzymes convert methylphenidate to the inactive metabolite ritalinic acid which is mainly (~90%) excreted in the urine.22 Immediate-release methylphenidate has a duration of action of three to five hours.22 The duration of action of modified-release formulations ranges from 8 – 12 hours, however, the pharmacokinetic profiles differ between formulation types; therefore, they are not generally considered interchangeable.22 External factors, such as taking medicines with food, can also influence methylphenidate absorption, e.g. taking Rubifen SR with a high-fat meal increases the rate of absorption (but does not affect the total amount absorbed).23
Different delivery technologies are used in modified-release formulations of methylphenidate which means that although brands are bioequivalent (considered to have the same clinical effect), variations in response and duration of effect may occur for individual patients changing between these medicines.24 Patient-specific factors may also influence therapeutic effects, e.g. preconceptions about generic medicines or anxiety about treatment changes.25
Dexamfetamine sulfate (dextroamphetamine sulphate) is an immediate-release psychostimulant medicine that is rapidly absorbed from the gastrointestinal tract and distributed throughout the body.27 It is metabolised hepatically to the pharmacologically active metabolite 4‐hydroxyamphetamine or phenylacetone; dexamfetamine, and its metabolites, are excreted renally.27 Changes in urinary pH influence dexamfetamine plasma half-life; alkaline urine slows excretion while urinary acidifying compounds increase excretion rates.27 Modified-release dexamfetamine formulations are available overseas but none are currently approved or funded in New Zealand.
Lisdexamfetamine dimesilate, a pharmacologically inactive compound (i.e. a prodrug), is rapidly absorbed into the bloodstream from the small intestine following oral administration and hydrolysed to dexamfetamine (the active form) and L-lysine in red blood cells.27, 28 The conversion process takes approximately 90 minutes; this means that there is less potential for misuse of this medicine compared to immediate-release dexamfetamine.28 The time to maximum therapeutic effect for lisdexamfetamine is delayed by approximately one hour compared to dexamfetamine.27 Linear dose equivalence for lisdexamfetamine and dexamfetamine cannot be established due to the differences in metabolism.29
Table 2. Overview of funded psychostimulant medicines available for managing ADHD in adults and children aged six years and over as of 1st February, 2026.1, 2, 23, 25, 30, 35–43 N.B. The data in this table are compiled from the New Zealand Formulary (and New Zealand Formulary for Children), the ADHD Prescribing Guide for Australian Healthcare Professionals, New Zealand data sheets and expert opinion, and should only be used as guide.
Open table in pop-up window
|
| Medicine |
Methylphenidate |
Dexamfetamine
(unapproved indication in adults) |
Lisdexamfetamine |
| Special Authority application form: |
SA2590
(Rubifen LA will be included on this form if funded) |
SA2591 |
SA2587 |
SA2588 |
| Brand: |
Ritalin |
Rubifen |
Rubifen SR |
Methylphenidate ER - Teva |
Methylphenidate Sandoz XR (provisionally approved until September, 2027) |
Rubifen LA
(proposed to be funded from 1st July, 2026) |
Ritalin LA |
Concerta |
Dexamfetamine (Noumed) |
Vyvanse |
| Medicine type: |
Innovator |
Generic (of Ritalin) |
Generic (of Ritalin SR) |
Generic (of Concerta) |
Generic (of Concerta) |
Generic (of Ritalin LA) |
Innovator |
Innovator |
Generic |
Innovator |
| Formulation and strengths available: |
Tablet
10 mg |
Tablet
5 mg
10 mg
20 mg |
Modified-release tablet
20 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Modified-release capsule
10 mg
20 mg
30 mg
40 mg
60 mg |
Modified-release capsule*
10 mg
20 mg
30 mg
40 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Tablet
5 mg |
Capsule†
30 mg
50 mg
70 mg |
| Onset of action: |
20 minutes – 1 hour |
20 minutes – 1 hour |
1 – 2 hours |
1 – 2 hours |
1 – 2 hours |
1 – 2 hours‡ |
1 – 2 hours |
1 – 2 hours |
20 minutes – 1 hour |
60 – 90 minutes |
| Duration of action: |
3 – 5 hours |
3 – 5 hours |
Up to 8 hours |
Up to 12 hours |
Up to 12 hours |
6 – 8 hours |
6 – 8 hours |
Up to 12 hours |
4 – 6 hours |
8 – 14 hours |
| Contraindications (for the full list of contraindications, see the NZF or New Zealand data sheets): |
- Acute severe depression, uncontrolled bipolar disorder, suicidal ideation or psychosis – patient should be stabilised first (involving specialist care)
- Anorexia nervosa
- Uncontrolled substance dependence or alcohol misuse (including in family and caregivers), however, expert opinion is that treatment could be carefully considered as part of a complete treatment plan if this is well-controlled or in remission (under specialist supervision)
- Hyperthyroidism
- Pre-existing cardiovascular disease, e.g. advanced arteriosclerosis, heart failure, myocardial infarction, arterial occlusive disease, cardiomyopathy, moderate to severe hypertension, arrhythmias, structural cardiac abnormalities
- Cerebrovascular disorders including aneurysm, vasculitis, vascular abnormalities, stroke — due to effects on heart rate and blood pressure
- Acute angle-closure glaucoma
- Hypersensitivity to the active ingredient
Note: Modified-release tablets should not be prescribed to patients with dysphagia or restricted size of gastro-intestinal lumen, e.g. gastric bypass |
|
|
| Cautions (for the full list of cautions, see the NZF or New Zealand data sheets): |
Psychostimulant medicines may precipitate or worsen:
- Anxiety or agitation
- Epilepsy
- Mild hypertension (or other cardiovascular conditions that may be compromised by increases in blood pressure or heart rate)
- Psychiatric disorders, e.g. bipolar disorder, or aggressive behaviour
- Tics – an association between psychostimulant medicine and development of tics was previously established but evidence now suggests this effect is no greater than placebo (or tic-specific treatments).44 The development of tics may be related to the emotions surrounding initiating treatment and the fluctuating nature of tics (caution is also recommended if the patient has a family history of Tourette syndrome).
Caution is also recommended when prescribing psychostimulants to patients with:
- Past history of disordered eating or difficulty maintaining weight
- Past history of substance or alcohol dependence or misuse (including in family and caregivers)
|
Key interactions
(for the full list of interactions, see the NZF or New Zealand data sheets): |
- Monoamine oxidase inhibitors, e.g. tranylcypromine, moclobemide
- Concurrent use can lead to hypertensive crisis – do not use within 14 days
- Tricyclic antidepressants
- Concurrent use may increase plasma concentrations of tricyclic antidepressants and increase the risk of adverse effects, e.g. cardiovascular effects
- Selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs)
- Concurrent use may increase the risk of serotonin syndrome
- Medicines that influence blood pressure
- Blood pressure control may be affected in patients taking antihypertensives, e.g. amlodipine, losartan, quinapril
- Increased risk of hypertension with concurrent use of pseudoephedrine, phentermine
|
- Opioids, e.g. codeine, tramadol
- May increase analgesic effect
- Antipsychotics, e.g. chlorpromazine
- May reduce the effect of amfetamines
|
Key adverse effects
(for the full list of adverse effects, see the NZF or New Zealand data sheets): |
Common:
- Insomnia
- Nausea and stomach discomfort, reduced appetite
- Sweating
- Hypertension, tachycardia and palpitations
- Anxiety, agitation, depressed mood, dysphoria, headache, irritability, tics – establishing baseline symptoms before initiating treatment is critical to distinguish adverse effects of psychostimulant treatment
Rare:
- Hepatic dysfunction
- Angina, myocardial infarction, supraventricular tachycardia
- Psychosis
- Cerebrovascular disorders, e.g. vasculitis, haemorrhage, cerebral arteritis, and vascular occlusion
- Tourette syndrome (in predisposed individuals)
- Seizures
- Angle-closure glaucoma
|
| Starting dose: |
5 mg, two to three times daily |
5 mg, two to three times daily |
20 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
20 mg, once daily in the morning |
20 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
2.5 mg, two to three times daily |
20 – 30 mg, once daily in the morning |
| Suggested titration based on response: |
Increase dose by 5 mg, weekly |
Increase dose by 5 mg, weekly |
Increase dose by 20 mg, weekly |
Increase dose by 18 mg, weekly |
Increase dose by 18 mg, weekly |
Increase dose by 20 mg, weekly (in adults) |
Increase dose by 20 mg, weekly (in adults) |
Increase dose by 18 mg, weekly |
Increase dose by 5 mg, weekly |
Increase dose by 10 – 20 mg, at a minimum of weekly intervals |
| Maximum dose: Discussion with, or referral to, a psychiatrist recommended if higher doses are being considered |
60 mg, divided over two to three doses, daily |
60 mg, divided over two to three doses, daily |
60 mg, once, daily |
72 mg, once daily |
72 mg, once daily |
80 mg, once daily |
80 mg, once daily |
72 mg, once daily |
20 mg, divided over two to three doses, daily, however, doses of 30 mg per day are commonly used
(up to 40 mg/day required in some children) |
70 mg, daily |
| Excipients with known effects or derived from animal sources: |
Gelatine, gluten, lactose |
None |
Lactose |
Lactose |
Lactose |
Gelatine |
Gelatine |
Lactose |
Lactose |
Gelatine |
| Comments: |
Can be taken with or without food |
Can be taken with or without food |
Should be taken with food – onset of action is faster when taken with a high-fat meal
Fewer fluctuations in plasma concentrations compared to multiple doses of immediate release methylphenidate |
Can be taken with or without food |
Can be taken with or without food |
Can be taken with or without food
Capsules can be opened, and beads can be sprinkled on cold food if required for administration |
Can be taken with or without food
Capsules can be opened, and beads can be sprinkled on cold food if required for administration |
Can be taken with or without food |
Can be taken with or without food |
Capsules can be opened and contents dissolved in water or juice, or mixed through food if required for administration |
* 60 mg Ritalin LA capsules are also available but not funded
† 20 mg, 40 mg and 60 mg lisdexamfetamine capsules are also available but not funded
‡ Rubifen LA is a generic of Ritalin LA and assumed to have the same onset and duration of action as the innovator
Best Practice Tip: Caution is recommended when switching between brands of methylphenidate and close monitoring and more frequent follow-up is advised. Generic medicines have demonstrated comparable bioavailability to innovator medicines and are considered bioequivalent by Medsafe.25 However, some patients may experience differences in effect when switching between innovator and generic medicines due to the medicine’s therapeutic index, variation in pharmacokinetics or patient populations, or patient factors, e.g. the nocebo effect.25 Clinicians with experience in prescribing these medicines have observed that differences in effect can occur when switching between formulations of methylphenidate, which is also noted in the New Zealand Formulary.30
Addressing inequities in accessing psychostimulant medicines for ADHD
Persisting health inequities experienced by Māori and Pacific peoples, and those living in areas of higher socioeconomic deprivation, living with disability or living rurally are widely acknowledged.45–48 It is likely that these established inequities further contribute to the barriers and challenges in diagnosing ADHD and accessing treatment, however, focused research in this area is limited.
A snapshot of psychostimulant medicine dispensing in New Zealand
By ethnicity. There is substantial variation between ethnicities in the number of people dispensed psychostimulant medicines (Figure 3). In 2020, rates of psychostimulant medicines dispensing were similar among European/Other (7.5 patients per 1,000 registered patients) and Māori (7.2 patients per 1,000 registered patients). Lower rates were observed for people of Pacific (2.3 patients per 1,000 registered patients) and Asian ethnicity (1.5 patients per 1,000 registered patients), suggesting underdiagnosis of ADHD in these groups or different approaches to treatment, e.g. preferring non-pharmacological management strategies.
Over time, the dispensing rate for European/Other (18.8 patients per 1,000 registered patients in 2024) has increased more rapidly compared to Māori (14.5 patients per 1,000 registered patients), Pacific (4.5 patients per 1,000 registered patients) and Asian (3.8 patients per 1,000 registered patients). This widening gap provides evidence of barriers to receiving an ADHD diagnosis and treatment for some groups. It has been proposed that reasons for the disparity for Māori may include difficulties in accessing medicines, e.g. medicines co-payments, cost of a primary care consultation, getting time off work, geographical distance for those who live rurally, and cultural beliefs regarding the use of medicines in the treatment of ADHD.49–51 There is limited research into the dispensing gap observed for people of Pacific and Asian ethnicities, however, medicine access (particularly for Pacific peoples) and cultural barriers are likely also contributors.
By deprivation status. Psychostimulant medicine dispensing for ADHD is highest for people living in the least deprived socioeconomic quintile (quintile 1) and dispensing rates decrease as deprivation increases (Figure 4). The cost of self-funding a private ADHD assessment, particularly for adults, is likely a key contributor to these disparities.
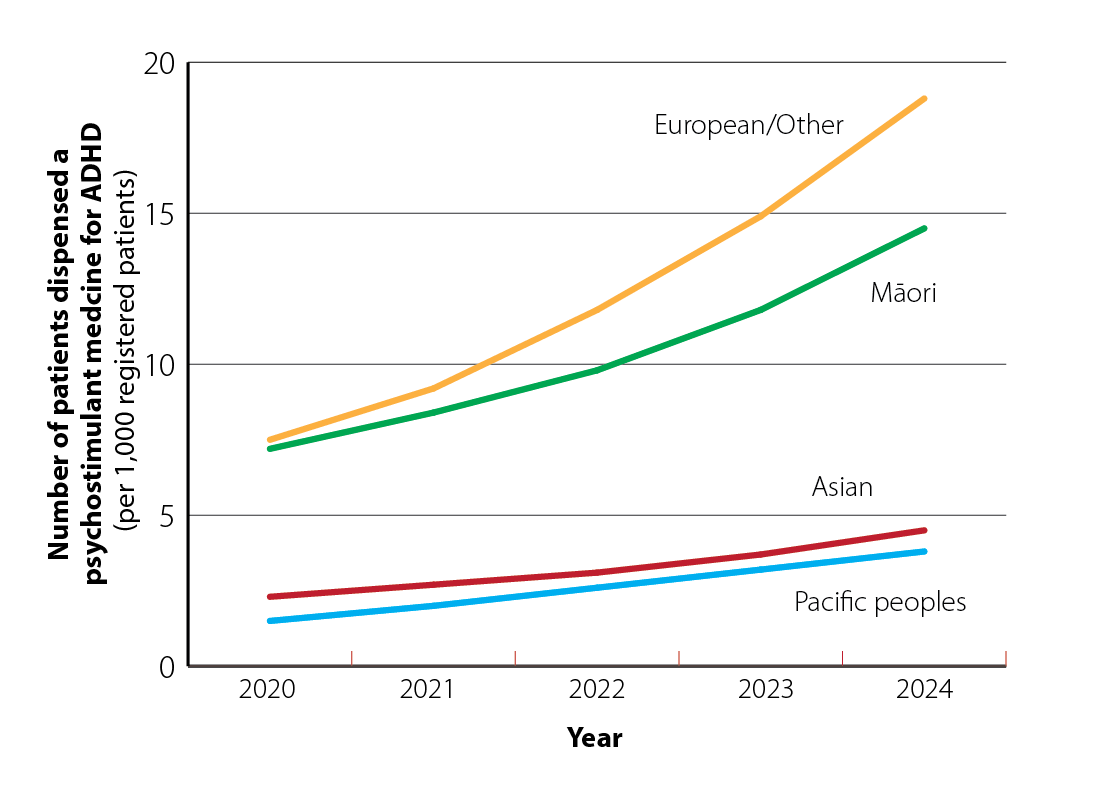
Figure 3. Number of patients dispensed psychostimulant medicines by ethnicity (per 1,000 registered patients) from 2020 – 2024. Data obtained from Ministry of Health, Manatū Hauora, Pharmaceutical Claims Collection, 2025.
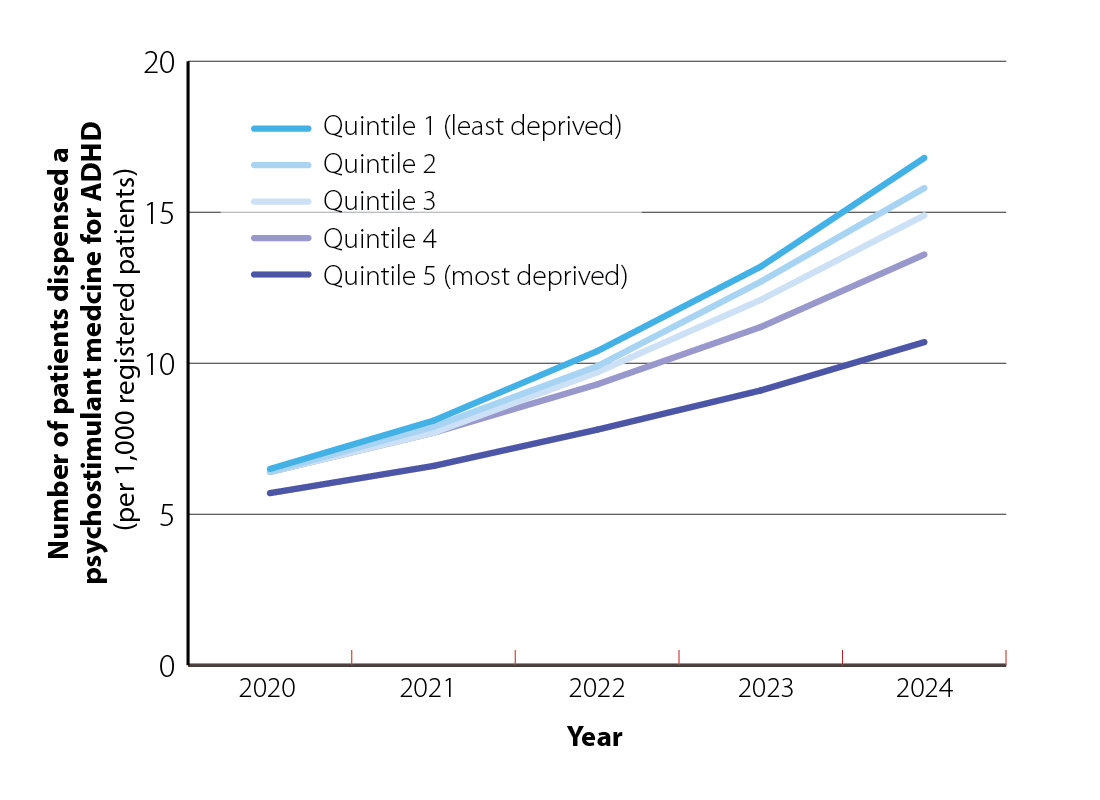
Figure 4. Number of patients dispensed psychostimulant medicines in New Zealand by socioeconomic quintile (per 1,000 registered patients) from 2020 – 2024. Data obtained from Ministry of Health, Manatū Hauora, Pharmaceutical Claims Collection, 2025.
By region. Capital and Coast had the highest rate of dispensing of psychostimulant medicines for ADHD in 2024; a 180% increase from 2020 (Figure 5). Other regions with high rates of ADHD medicine dispensing are typically larger centres with major tertiary hospitals, e.g. Auckland, Waitematā and Hutt Valley. ADHD medicine dispensing was lower than average in Northland, Lakes, Tairāwhiti and South Canterbury. Challenges with accessing secondary care health services in New Zealand are well established.52 Poorer access to secondary care, e.g. paediatricians or psychiatrists, in these regions, compared to major centres could explain some of the observed regional variation.
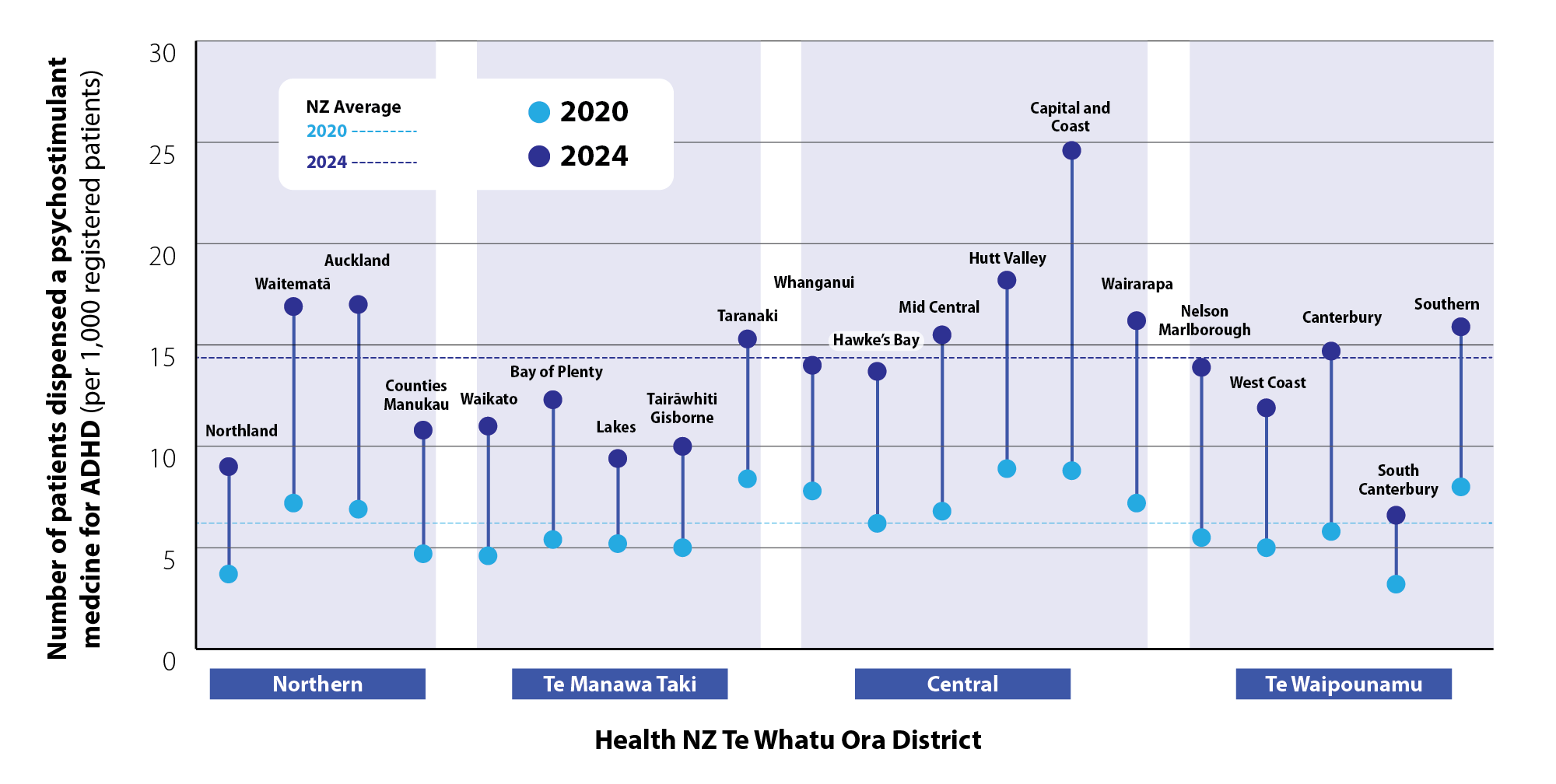
Figure 5. Number of patients dispensed psychostimulant medicines in New Zealand by region (per 1,000 registered patients) in 2020 and 2024. Data obtained from Ministry of Health, Manatū Hauora, Pharmaceutical Claims Collection, 2025.
Prioritise culturally appropriate ADHD diagnosis and management
A culturally appropriate approach to the diagnosis and management of ADHD in New Zealand is critical. Specific research in this area is lacking, however, it is likely that Māori and Pacific peoples hold perspectives and attitudes to neurodiversity and ADHD that fall outside the conventional biomedical model, as for example is seen in mental health. Māori and Pacific peoples experience higher rates of mental health distress than the overall New Zealand population, while also having higher unmet needs for professional mental health services.53 Misalignment between Māori and Pacific perspectives and attitudes to mental health conditions and Western health models has been cited as a potential contributor to these disparities, along with colonisation and systemic racism, and drive the need for culturally appropriate mental health services in New Zealand.54–56 Given inequities in ADHD medicine dispensing have been identified (see Figure 3), primary care prescribers who choose to offer ADHD services must endeavour to do so in a way that acknowledges and respects different worldviews and takes cultural considerations into account.57
Removing barriers to accessing psychostimulant medicines in primary care
Changes at a health system level are required to address many of the factors contributing to the barriers to ADHD treatment. The widening of access to psychostimulant medicines for ADHD in the community could provide opportunities that some people may not have had access to previously and lay the foundation to build patient-centred community-based models of care.
At an individual level, clinicians must be able to recognise patients in whom screening for ADHD is appropriate and where possible, support them to undergo an assessment, which may result in a diagnosis. It is well established that people with untreated ADHD have lower life expectancy and experience higher rates of adverse functional outcomes.1, 58 Positive effects of psychostimulant treatment on quality of life, suicidal behaviours, substance misuse, transport accidents, and criminality are beginning to emerge in addition to improvements in core ADHD symptoms.33, 34, 58
Practical steps for primary care prescribers
Once a patient has received a diagnosis of ADHD, it is important to actively involve the patient and their family/whānau when establishing a treatment plan, and ensure that cultural considerations are incorporated.57 There is limited evidence on how to approach this in the context of ADHD management in primary care, but practical suggestions from other fields can be extrapolated:1, 2, 57, 59–61
- Acknowledge the significant inequities for Māori, Pacific peoples and other groups in the New Zealand health system
- Take time to build relationships with new patients and their family/whānau, e.g. whakawhanaungatanga
- Use cultural health models to guide clinical interactions where appropriate, e.g. Te Whare Tapa Whā model of Māori health, Meihana Model framework, Hui Process, Fonofale model of Pacific health (Pan-Pacific), Fonua Ola (Tonga)
- Neuro-affirming*, culturally appropriate communication is vital;
- Discuss the possible advantages and strengths conferred by having ADHD, such as intelligence, creativity, adventurousness, humour and ability to develop compensatory mechanisms
- De-emphasise the “disorder” aspect of the diagnosis and reassure that challenges can be overcome with the appropriate treatments, supports and accommodations, e.g. use the analogy of getting glasses for short-sightedness
- Incorporate cultural concepts and narratives when discussing the symptoms, diagnosis and management of ADHD, e.g. using the behaviour of the pīwakawaka (New Zealand fantail), such as curiosity, constant movement and adaptability, as a metaphor for ADHD characteristics; using the term “aroreretini” (the te reo Māori word for ADHD which translates to “attention goes to many things”)
- Support people to incorporate their personal beliefs and knowledge systems into their management plans, e.g. Te Ao Māori (Māori world view)
- Ensure information on psychostimulant medicine choices, including medicines funding (Special Authorities) and regulatory issues (Controlled Drug restrictions) is provided in a format that is appropriate for the health literacy level of the patient and their whānau. Check that the information provided has been understood.
- Allow the patient and their whānau time to decide on which treatment is appropriate for them and help them to navigate this process
* A neuro-affirming approach acknowledges and accepts differences in neurological processing and validates and empowers neurodivergence, in contrast to focusing on deficits. It is collaborative, adaptable and encompasses shared decision-making.
A high level of diagnostic confidence is required when prescribing psychostimulant medicines.62 The decision to trial pharmacological treatment for ADHD must be made in conjunction with the patient (and their caregivers where appropriate) as part of a shared decision-making process.1 As when prescribing any medicine, discussion should cover the potential benefits and adverse effects of treatment, different medicine options and dosing regimens available, and any patient factors that may influence treatment adherence or safety, e.g. co-morbid conditions, family and social support, previous substance use disorder (see: “Prepare patients for starting psychostimulant medicines”).
Pre-treatment screening and investigations
As part of minimum screening requirements prior to prescribing psychostimulant medicines to a patient in primary care:
- Review medical history and any other currently prescribed medicines to identify cautions and rule out any contraindications (Table 2)1
- Record weight and conduct a cardiovascular assessment, including heart rate and blood pressure1
- Assess risk for substance misuse or diversion by the patient and their family or caregivers30, 63
Further cardiovascular screening considerations
The cardiovascular safety of psychostimulant medicines is a key consideration for prescribers (see: “Read the evidence on psychostimulant medicines and cardiovascular disease”). ADHD itself is also an independent risk factor for the development of cardiovascular disease in adults;64 patients should be informed of the increased risk of cardiovascular disease associated with both ADHD and psychostimulant medicines, as well as factoring in their own current baseline cardiovascular health when making decisions regarding treatment. In practice, most people with ADHD will have a lower absolute risk of cardiovascular events and primary care prescribers should use their clinical judgement when considering how initiating psychostimulants may increase this risk on an individual level. An electrocardiogram (ECG), cardiologist opinion or referral for cardiology assessment may be appropriate in the following clinical situations:1
- Prescribed concurrent medicines that “may pose an increased cardiac risk”. Interpretation of this recommendation varies. Any medicine prescribed for a cardiac condition warrants discussion with a cardiologist. At minimum, an initial ECG is suggested for patients who regularly take any medicines with potential cardiovascular adverse effects, e.g. tricyclic antidepressants.
- Hypertension. Ideally, patients with any level of hypertension should have a baseline ECG.
- A history of congenital heart disease, cardiac surgery or sudden cardiac-related death in a first-degree relative aged under 40 years
- Shortness of breath or fainting on exertion
- Heart palpitations
- Suspected cardiac chest pain
- Heart murmur on examination
Read the evidence on psychostimulant medicines and cardiovascular disease
The cardiovascular safety of psychostimulant medicines remains a concern, despite the majority of people with ADHD generally having low absolute risk of cardiovascular events.65 A Swedish study involving more than 250,000 participants showed a small (10%) increase in the rate of cardiovascular events in the first six months of treatment among those with ADHD taking methylphenidate (median age at treatment initiation of 22 years), compared to participants without ADHD and not taking methylphenidate.66 Long-term use of medicines used to treat ADHD has also been associated with an increased risk of cardiovascular disease, particularly hypertension and atherosclerosis.67 However, these findings also suggest that medicine dose and duration of treatment may influence risk; a significant increase in cardiovascular disease risk was only observed in people who were prescribed a mean methylphenidate dose that was 1.5 times the “defined daily dose” of 30 mg/day, i.e. people needed to take doses of 45 mg/day to experience an effect.67 Both of these studies highlight the importance of considering the individual risks and benefits of psychostimulant medicines before initiating treatment.66, 67
Establish symptom and function baseline
Assess the patient’s ADHD symptoms and daily functioning, i.e. their effect on employment, education, sleep, to act as a baseline for dose titration and measuring treatment response; standard scales may be useful for this (see: “Scales for monitoring treatment response”).1, 63 The optimal dose for psychostimulant medicines is guided by improvements in core ADHD symptoms and daily functioning, and development of adverse effects.1
Best Practice Tip: Set individualised treatment goals in collaboration with the patient.2 Identify areas in which ADHD symptoms have the most significant impact on their life and determine realistic improvements, e.g. successful meeting of an important deadline at work.2 The Specific, Measurable, Attainable, Relevant and Timely (SMART) framework is often useful when setting treatment goals.2
Identifying and addressing co-morbidities is critical
Assess for (and manage) co-morbid mental health conditions before initiating psychostimulant medicines, and identify any substance use disorders or suicidal ideation, which may impact treatment decisions.1 People living with ADHD are at higher risk of anxiety, depression, bipolar disorder and substance use disorder.68 For patients already established on treatment for a mental health condition, e.g. anxiety, re-evaluate their current treatment plan, including non-pharmacological management and check for medicine interactions. In some cases, anxiety and depression may occur secondary to ADHD; effective treatment of core ADHD symptoms may result in overall improvement in anxiety and depression symptoms.1, 2 Consider screening for ADHD in patients with anxiety or depressive symptoms that do not respond to standard treatments.1
Psychostimulant medicines and eating disorders. Psychostimulant medicines are contraindicated in patients with uncontrolled anorexia nervosa due to their appetite suppression effects.30 Expert opinion is to generally avoid psychostimulant medicines if there are any concerns about the patient maintaining body weight during treatment, however, treatment could be considered on a case-by-case basis, e.g. psychostimulants may be appropriate in patients with co-existing anorexia nervosa who have been effectively treated, have ongoing support and are maintaining their goal weight. If treatment is initiated in a patient with a history of disordered eating, close monitoring and potentially slower dose titration is required.1 For advice on managing appetite suppression as an adverse effect of psychostimulant medicines, see: “Monitoring and follow-up of adults stabilised on psychostimulant medicines”.
ADHD and substance misuse
ADHD is an established risk factor for the development of substance misuse.1 Concurrent treatment for patients presenting with both ADHD and substance misuse is recommended.1, 2 Concern regarding prescribing psychostimulant medicines in patients with a history of substance misuse is understandable given their mechanism of action (increased dopaminergic activity).1 As such, prescribing psychostimulant medicines in patients with uncontrolled substance misuse should be avoided and treatment may need to initially focus on abstaining or reducing substance misuse.
Available evidence shows treatment of ADHD with psychostimulant medicines does not increase the risk of substance misuse, when compared to people with ADHD who do not receive treatment.1 Treatment of ADHD with psychostimulant medicines, in some cases, may reduce substance use disorder.1, 34 Prescribers should carefully consider whether extended-release or prodrug formulations of psychostimulant medicines could be included as a component of an overall treatment programme and occur under supervision of a psychiatrist or other relevant specialist. N.B. Non-psychostimulant medicines may have a role in this patient group.
Psychostimulant medicines during pregnancy and breast-feeding
Pregnancy and breast-feeding are not considered outright contraindications to the use of all psychostimulant medicines for ADHD but there is limited data on these situations.69 Recommendations for similar medicines may differ based on what studies have been carried out rather than a direct comparison of safety, e.g. dexamfetamine is contraindicated in pregnancy (and breast-feeding) due to potential risks shown in clinical studies, while there is an absence of data on the effects of lisdexamfetamine in pregnancy, hence caution is advised rather than a contraindication. Clinicians must therefore balance the possible benefits and risks to the mother (e.g. risk of untreated ADHD) and fetus/infant (e.g. adverse effects of medicines) when deciding whether to continue psychostimulant medicines during pregnancy and the post-partum period.70 Ideally, this decision should be made in conjunction with a psychiatrist or obstetrician.
Pregnancy
Methylphenidate and lisdexamfetamine can be cautiously considered in pregnancy if the benefits of treatment are considered to outweigh the risks.*30 There may be a small increase in the risk of cardiac malformations when taking methylphenidate (evidence for amfetamines is conflicting), but exposure to psychostimulant medicines in utero is not associated with an increased risk of neurodevelopmental disorders.42, 71, 72 Possible small increases in the risk of pre-eclampsia, preterm birth, spontaneous abortion and neonatal intensive care admission have also been reported in infants born to mothers who took methylphenidate during pregnancy, however, these findings are not consistent and may be subject to confounding factors.69
If methylphenidate or lisdexamfetamine are prescribed, fetal growth, blood pressure and weight gain should be assessed frequently throughout pregnancy.69 Close monitoring of the infant is also recommended after birth for irritability, difficulty sleeping and breathing problems.30
Dexamfetamine is contraindicated by the manufacturer in pregnancy due to a the potential risk of vasoconstriction and reduced blood supply to the placenta which may increase likelihood of premature delivery and low birth weight.30 Dexamfetamine use during pregnancy has also been associated with congenital malformations, however, data to support this includes illicit use (as opposed to supervised medical use).30 Therapeutic use of dexamfetamine appears to be low risk for congenital malformations.30
* New Zealand data sheets state that Ritalin and Vyvanse “should not be given to pregnant women unless the potential benefit outweighs the risk to the fetus”35, 37
Breast-feeding
The manufacturers of methylphenidate and lisdexamfetamine advise to avoid use while breast-feeding, while dexamfetamine is contraindicated by the manufacturer.30 The limited available data suggests methylphenidate is excreted in small amounts in breast milk; a relative infant dose of < 1% has been reported (doses < 10% are generally considered safe).69 There are no reports of adverse effects in infants whose mothers were taking methylphenidate during breast-feeding.69 Amfetamines are also excreted in breast milk but no significant adverse effects are expected in the short term (long-term data are not available).69
If the benefits outweigh the risks and a shared-decision is made to continue psychostimulant treatment during breast-feeding, monitor the infant for adverse effects, e.g. irritability, difficulty sleeping and feeding.69 Psychostimulant medicines may also impact milk production due to increased levels of dopamine inhibiting prolactin, especially if lactation has not yet been established.69
 Pregnancy should ideally be planned for females with ADHD.69 Some people may prefer to stop taking psychostimulant medicines during pregnancy. Offer a trial of a treatment holiday well in advance of a planned pregnancy to assess the possibility of stopping treatment.69 Increase focus on lifestyle changes and non-pharmacological management also.
Pregnancy should ideally be planned for females with ADHD.69 Some people may prefer to stop taking psychostimulant medicines during pregnancy. Offer a trial of a treatment holiday well in advance of a planned pregnancy to assess the possibility of stopping treatment.69 Increase focus on lifestyle changes and non-pharmacological management also.
Prepare patients for starting psychostimulant medicines
Treatment expectations often differ between prescribers and patients living with ADHD. It is important that realistic treatment outcomes are discussed.2 Patients should be aware that pharmacological treatment reduces symptoms but complete resolution is unlikely.1 Specific medicines and doses that effectively manage symptoms for some people may not be beneficial or tolerable for others;2 multiple trials of different medicines and dosing regimens may be required to find the optimum treatment. Medicines are most effective at reducing ADHD symptoms, but also are only one aspect of treatment; non-pharmacological interventions provide skills that further reduce the impact ADHD on patients’ lives.1 For example, focus may improve with psychostimulant medicines, however, it is up to the patient to decide where to direct that focus.
The need for pharmacological intervention can change throughout a person’s life (see: “Regularly revisit the need for treatment”).2 This may be due to life changes such as starting a new job in an environment where ADHD symptoms may be less challenging (or advantageous), or acute or long-term fluctuations in the severity of their ADHD symptoms, e.g. some females may experience more severe ADHD symptoms during the luteal phase of their menstrual cycle or during perimenopause (based on anecdotal reports).73 In some cases, treatment of ADHD with psychostimulant medicine enables more effective utilisation of non-pharmacologic strategies, e.g. cognitive behavioural therapy, optimising environment, routines. Patients can gain understanding of how to better manage their ADHD symptoms, in time allowing dose reduction, treatment holidays or targeted medicines use at key times in a person’s life or schedule, i.e. when the effect is required (see: “Tailoring the psychostimulant regimen over time”). A person who no longer requires pharmacological intervention for ADHD, still has ADHD, but the functional impairment has reduced to the point where a person manages it without medicine.
Advise patients to monitor their response to treatment and record both beneficial and adverse effects.1, 36 Adverse effects are more common when treatment is first started and following dose increases, but generally improve after a short period of consistent dosing.2 Explain what the patient may experience if the dose is too high, e.g. feeling “wired” or irritable, excessive (or difficulty shifting) focus, restricted affect (“zombie effect”),2 and what to do if this occurs, i.e. reduce their dose or contact their prescriber.
People living with ADHD often find taking medicine as prescribed challenging.1 Strategies to promote medicine adherence include:1, 63
- Provide clear dosing instructions, e.g. work with the patient to create a dosing schedule that is optimised to their daily requirements and present the information in a format that is most appropriate for them such as written instructions, weekly dosing table or a dosing calendar
- Aim for a once daily dosing regimen, where possible
- Discuss ways to incorporate taking medicines into the patient’s daily routine to prevent missed doses, e.g. storing medicines near their toothbrush, using a pill dispenser, setting reminders using phone applications, alarms or diary notes to take doses, collect repeat prescriptions and attend follow-up appointments. Clinicians could also place an appropriate recall for regular follow-up in the patient’s clinical record.
- Share information about joining peer support groups, e.g. ADHD New Zealand, social media groups
Information for patients (and clinicians) about keeping a diary to monitor their ADHD treatment response, including a checklist for what to include is available here. A patient form to guide discussion with a prescriber about adverse effects is available here.
Prescribe the most appropriate formulation based on the clinical situation, patient preference and medicine availability.1 Generally, patients should be initiated on the lowest available dose of the chosen formulation and titrated up slowly. However, titrating psychostimulant medicines is often a trial and error process and must be individualised depending on the patient’s daily requirements and therapeutic response. There is no evidence to support one protocol over another; primary care clinicians with previous experience initiating psychostimulant medicines under the supervision of a psychiatrist may already have an established process they follow. The following information is intended as a guide and is based on published guidelines and expert clinical opinion.
 Discussion with a psychiatrist is recommended if there are any queries or concerns prior to initiation of psychostimulant medicines. Primary care clinicians should have a low threshold for consulting with a psychiatrist if initiating psychostimulant medicines in patients with concomitant mental health conditions, e.g. schizophrenia or bipolar disorder, personality disorders, eating disorders, post-traumatic stress disorder, or substance misuse. Patients with complex needs or high-risk clinical features should not be managed in primary care; referral to psychiatric services is recommended.62
Discussion with a psychiatrist is recommended if there are any queries or concerns prior to initiation of psychostimulant medicines. Primary care clinicians should have a low threshold for consulting with a psychiatrist if initiating psychostimulant medicines in patients with concomitant mental health conditions, e.g. schizophrenia or bipolar disorder, personality disorders, eating disorders, post-traumatic stress disorder, or substance misuse. Patients with complex needs or high-risk clinical features should not be managed in primary care; referral to psychiatric services is recommended.62
Choosing the right medicine – immediate- or modified-release?
Immediate-release formulations of methylphenidate and dexamfetamine are useful when initiating treatment, particularly in children, as they allow for simple dose titration. However, the fast onset and short duration of action may present problems for some people. Most people with ADHD will require several doses of an immediate-release formulation to manage their symptoms throughout the day, with the number of doses depending on their symptom severity and cognitive requirements.22 Multiple daily dosing adds medicine regimen complexity and can lead to poor treatment adherence, dosing errors, may increase the risk of adverse effects due to fluctuations in blood concentrations or rebound ADHD symptoms at the end of the medicines’ duration of action (rapid “weaning off” effect).2, 22 Social stigma and negative beliefs relating to ADHD and psychostimulant medicines may also affect people who need to take their medicine at work or school, and must be considered.22
Modified-release formulations of methylphenidate, and lisdexamfetamine, have several advantages over immediate-release psychostimulants.1 Single-daily dosing is often more convenient for people and can improve adherence.1 The need to take multiple doses at work or school is also reduced. Modified-release formulations lessen or avoid sharp blood concentration spikes and drop-offs, reducing the potential for adverse effects and rebound ADHD symptoms when the treatment wears off.1 Furthermore, modified-release formulations, particularly Concerta, and lisdexamfetamine, are less likely to be misused or abused because the immediate-release component of the overall dose is often smaller and the delivery technology prevents tampering/makes other delivery methods (e.g. insufflation [snorting], injection) ineffective.1
Consider initiating a longer-acting medicine formulation in adults who require treatment to cover a longer duration of their day, do not want to take multiple daily doses or have found adherence to previous treatment regimens for other conditions challenging, or if there is potential for abuse, misuse or diversion.1, 2
Lisdexamfetamine would typically be considered after a trial of another option, however, Special Authority criteria enables it to be used first-line if the patient is unable to access other psychostimulant medicines due to supply issues or the prescriber has concerns regarding diversion or abuse of immediate-release medicines.
Initial monitoring during dose titration
Patients are typically initiated on the lowest available dose of the chosen formulation, with the dose slowly increased in steps depending on treatment response and the development of any adverse effects.1, 2 Frequent monitoring is necessary during the titration process.2 Virtual follow-up, e.g. phone or video consultation, can be useful to check in with the patient, with an in-person clinical review occurring within two to four weeks of starting treatment.1, 2, 57 Prescribers should use their clinical judgement when determining monitoring frequency in patients with co-existing conditions. Slower dose titration, more frequent in-person monitoring or discussion with a psychiatrist may be appropriate in patients with:1
- Other neurodevelopmental disorders e.g. autism spectrum disorder, tic disorders, intellectual disability
- Concomitant mental health conditions, e.g. anxiety disorders, depression, schizophrenia or bipolar disorder, personality disorders, eating disorders, post-traumatic stress disorder, substance misuse
- Any other condition that may be worsened while taking psychostimulant medicines, e.g. cardiovascular disease (including hypertension), epilepsy
Table 3 provides suggested starting doses and increments for dose increases for the different funded psychostimulant medicines available in New Zealand.
Scales for monitoring treatment response
Validated symptom questionnaires (diagnostic/screening tools) can be used to establish a baseline for ADHD symptoms, assess symptom improvement and measure treatment response, including personal treatment goals.2, 63
Commonly used questionnaires include:
- Adults
- Adult ADHD Self-Report Scale – an 18-item questionnaire rating symptoms on a five-point scale. This tool takes approximately five minutes to complete.
- Children
In children, questionnaires will usually need to be completed by parents/caregivers. Additional information from teachers is valuable when managing children living with ADHD as treatment effects of psychostimulants are typically beneficial for school performance and may not be fully realised by parents or caregivers. Suggest that parents liaise with the child’s school and arrange for an appropriate questionnaire to be completed by the child’s teacher and brought to the follow-up appointment.
The Australian Evidence-Based Clinical Practice Guideline For Attention Deficit Hyperactivity Disorder (2022) contains a more exhaustive list of questionnaires (Box 1) as well as principles for monitoring treatment responses.
Finding the right daily dosing regimen for the individual
The optimal dose of a psychostimulant medicine reduces core ADHD symptoms and improves functional outcomes with minimal adverse effects.1 There is substantial inter-person variation in psychostimulant doses; expert opinion is that a treatment response is typically expected at doses of 0.5 – 1 mg/kg for methylphenidate. However, some adult patients report adequate symptom improvement when taking a total daily dose of only 20 mg methylphenidate. This highlights the importance of an individualised approach to treatment and dose titration.
At every follow-up, review:
- Are the patient’s core ADHD symptoms well controlled, i.e. low scores on symptom scales?
- Does symptom control vary throughout the day (or from day to day) and does this variation present challenges for the patient?
- Is the duration of action correct for the individual needs of that patient?
- Are there any adverse effects that warrant a dose reduction?
Increase or decrease the dose of psychostimulant medicine based on the responses to these questions.
Switching to a modified-release formulation
Patients initiated on immediate-release formulations of methylphenidate may want to move to a modified-release formulation. Once the patient’s total daily dose is established in milligrams, they could be switched to the equivalent dose of a modified-release formulation, if appropriate. Patients initiated on dexamfetamine who want to switch to lisdexamfetamine, and vice versa, will need to start on the lowest dose and titrate up again as they are not dose equivalent (Table 3).29
Tailoring the psychostimulant regimen over time
Once the appropriate formulation and optimal dose have been established, there may be opportunities to further adjust the patient’s dosing schedule to ensure treatment is targeted to key periods in which the highest functional demands exist. Ideally, clinicians should continue to work with patients to formulate an individualised plan to suit their needs.2 In some cases, it may be appropriate for patients to be given the autonomy to adjust their doses as required.
Combining methylphenidate formulations may be beneficial
Immediate-release and modified-release formulations can be used at different times throughout the day to optimise the duration of action for the individual patient.1 Ideally, the timing of doses should result in effective treatment of symptoms during the periods in which patients have their highest cognitive requirements (which may not necessarily just be related to work/study). Immediate-release methylphenidate doses can be used to provide symptom control before the effects of a modified-release formulation become apparent or later in the day when the effects of the modified-release formulation have decreased. Timing is important: the immediate-release dose does not need to be withheld until the end of the expected duration of the modified-release dose (it can be taken as the effect is waning), but it should also not be taken too close to the expected peak effects of the long-acting dose (as this may cause adverse effects). Extra dosing may also only be needed during certain times of extra cognitive load, rather than every day.
Changing from continuous to scheduled dosing
It is important when initiating psychostimulant medicines, that doses are taken consistently (i.e. every day) to observe effects on all areas of the patient’s life. However, once an optimal dose is achieved, supportive non-pharmacological strategies have been implemented and the patient has a better understanding of how the psychostimulant medicine affects them (and those around them) day-to-day, a discussion could take place regarding flexible dosing regimens. For example, some people may prefer to not take their psychostimulant medicine during the weekends or to take reduced doses on certain days to avoid adverse effects.6 Conversely, many people experience benefit from psychostimulant medicines across all aspects of their lives and therefore, favour continuous dosing. These differences further highlight the importance of individualised medicine regimens and the need to work with patients to optimise treatment.
Table 3. Example initial dosing regimens for funded psychostimulant medicines in an adult patient in a primary care setting. N.B. This table is intended as a guide and starting doses are conservative; prescribers with more experience titrating psychostimulant medicines may be more confident starting at higher doses or increasing doses more quickly (see prescriber notes). The optimal dose varies substantially between individuals, and the titration process may take months in some cases.
Prescriber notes:
- Effective titration and follow-up when initiating psychostimulant medicines is critical and may influence long-term efficacy and treatment adherence
- Treatment effects are usually evident very early after a dose change, i.e. the day of the dose increase, however, a delay of up to one week before increasing to the next dose is suggested to ensure response is not related to other life changes occurring at the same time
- Depending on clinician confidence, shorter periods between dose increases may be appropriate, e.g. many psychiatrists favour increasing the dose every three to four days (if well tolerated) or starting at higher doses (a total daily dose of 10 – 20 mg immediate-release methylphenidate in a healthy adult with no cautions or contraindications)
|
- Phone consultations can be useful for follow-up during dose titration, but an in-person clinical review should occur within two to four weeks of starting treatment
- The optimal dose of a psychostimulant medicine reduces core ADHD symptoms and improves functional outcomes with minimal adverse effects. Ideally this is the lowest effective dose and with regular review, is generally apparent within eight weeks.
- Avoid increasing the dose when target symptoms are responding well and adverse effects are not present or mild, i.e. stopping dose titration after two or three weeks if an acceptable dose has been reached
- A reduction in symptom control with dose increase indicates the dose may be too high and warrants a dose decrease
|
| Immediate-release methylphenidate, e.g. Rubifen, Ritalin |
| Week |
Morning |
Midday |
| 1 |
5 mg |
5 mg |
| 2 |
10 mg |
5 mg |
| 3 |
15 mg |
5 mg |
| 4 |
20 mg |
5 mg |
| 5 |
25 mg |
5 mg |
| Maximum daily dose |
60 mg (in two to three doses) |
|
| Modified-release methylphenidate (up to eight hours duration of action), e.g. Rubifen SR, Ritalin LA (or Rubifen LA: proposed to be funded) |
| 1 |
20 mg |
– |
| 2 |
40 mg |
– |
| 3 |
60 mg |
– |
| Maximum daily dose |
80 mg, daily (60 mg for Rubifen SR) |
|
| Modified-release methylphenidate (up to 12 hours duration of action), e.g. Concerta, Methylphenidate ER - Teva, Methylphenidate Sandoz XR |
| 1 |
18 mg |
– |
| 2 |
36 mg |
– |
| 3 |
54 mg |
– |
| Maximum daily dose |
72 mg, once daily |
|
| Dexamfetamine (a single dose of dexamfetamine in the morning may provide sufficient effect for some people as the duration of action can be up to six hours) |
| 1 |
5 mg |
– |
| 2 |
5 mg |
5 mg |
| 3 |
10 mg |
5 mg |
| 4 |
10 mg |
10 mg |
| Maximum daily dose |
20 mg (in divided doses), however, doses up to 30 mg/day are commonly used in clinical practice |
| Lisdexamfetamine* |
|
|
| 1 |
30 mg |
– |
| 2 |
50 mg |
– |
| 3 |
70 mg |
– |
| Maximum daily dose |
70 mg, once daily |
|
| If adverse effects develop, advise patient to return to the previous dose where symptoms were controlled and adverse effects were not present. Continue at this dose until the next in-person follow-up. |
* 20 mg, 40 mg and 60 mg capsules are available (but not funded) and can be used for a more gradual titration in patients who experience adverse effects and are able to meet the cost of self-funding
Prescription considerations for psychostimulant medicines
Prescribe an adequate quantity of medicine to last until the patient’s next in-person follow-up/monitoring appointment, i.e. taking into account scheduled dose increases. Include clear instructions for dosing titrations, and where possible, provide separate written information, e.g. patient information handouts. Remind patients that prescriptions for psychostimulant medicines can only be dispensed within seven days of being electronically generated (or the date of signing of the prescription) and the maximum quantity they can be dispensed from the pharmacy at one time is enough for one month of treatment (with two repeats).74, 75 N.B. Psychostimulant medicines are classified as Class B Controlled Drugs under the Misuse of Drugs Regulations 1977 and cannot be prescribed in quantities of more than what is sufficient for three months of treatment, i.e. not eligible for extended prescribing.74
Navigating Special Authorities for psychostimulant medicines
Pharmac uses Special Authorities as a targeting tool to ensure medicines are funded for people who are most likely to benefit and to provide effective medicines budget management. There are four separate Special Authorities for psychostimulant medicines in New Zealand; Special Authority applications can be made by any relevant practitioner.75
For methylphenidate:
- SA2590 applies to first-line or lower-cost brands: Ritalin, Rubifen SR, Methylphenidate ER – Teva, Methylphenidate Sandoz XR (Rubifen LA is proposed to be listed on this Special Authority from 1st July, 2026)
- SA2591 applies to second-line or higher-cost brands: Ritalin LA, Concerta
For amfetamines:
- SA2587 applies to dexamfetamine sulfate
- SA2588 applies to lisdexamfetamine dimesilate which is funded as a second-line option unless there are supply issues or concerns regarding diversion or abuse
The development of these Special Authorities was based on clinical advice that most people respond well to the more cost-effective options, while managing funding and ensuring access to alternatives when needed.
To mitigate ongoing supply shortages, the usual targeting rules have been temporarily suspended (i.e. a first-line option does not need to be trialled before accessing a second-line brand), and in early 2025, people with an existing methylphenidate Special Authority number were given access to the other Special Authority number.9 As a result, most people receiving treatment for ADHD now have both SA2590 and SA2591 approval, and either may be used, depending on stock availability.
Patients who have recently started treatment may only have one Special Authority number. If their usual brand is out of stock and the alternative brand is not covered by their current Special Authority, the prescriber will need to apply for the second Special Authority number for the person to receive funded medicine.
It is expected the original targeting approach with preferred first- and second-line options will be reinstated once supply of these medicines stabilises.
Legal requirements when travelling overseas with psychostimulant medicines
 The maximum quantity of any psychostimulant medicine that the patient can take with them when they leave New Zealand, or be brought back into the country, is an amount sufficient for one month of treatment under the Misuse of Drugs Act 1975. The medicines should be kept in the labelled packaging from the pharmacy. Patients must travel with a copy of their prescription or a letter from the prescriber. Patients should also establish the legal status and requirements relating to carrying psychostimulant medicines in any countries they visit prior to departure.
The maximum quantity of any psychostimulant medicine that the patient can take with them when they leave New Zealand, or be brought back into the country, is an amount sufficient for one month of treatment under the Misuse of Drugs Act 1975. The medicines should be kept in the labelled packaging from the pharmacy. Patients must travel with a copy of their prescription or a letter from the prescriber. Patients should also establish the legal status and requirements relating to carrying psychostimulant medicines in any countries they visit prior to departure.
For further information on entering New Zealand with psychostimulant medicines, see: https://www.customs.govt.nz/travel-to-and-from-new-zealand/medicines
Arrange regular follow-up to assess treatment response and monitor for adverse effects once the patient is stabilised on the optimum dose of their psychostimulant medicine.1 The frequency should be determined by the patient’s clinical condition. A review every six months is appropriate for most patients with well-controlled mild to moderate ADHD symptoms.76 More frequent follow-up may be required in specific circumstances, e.g. severe ADHD symptoms, co-morbid psychiatric conditions, higher risk of adverse effects (or adverse effects have developed), medicines adherence or abuse/diversion concerns.1
Monitoring should include:
Evaluate ongoing improvement in ADHD symptoms.1 Use standard scales to measure therapeutic effect over time (see: “Scales for monitoring treatment response”).63
Measure weight, heart rate and blood pressure at every review.1 An ECG and laboratory investigations are not routinely recommended, but may be required if there is a clinical indication, e.g. liver function tests for suspected hepatic dysfunction.63
Assess for and address any adverse effects.
Sleep disturbances – discuss sleep hygiene techniques, e.g. having a consistent bedtime, avoiding devices at bedtime.3, 76 Establish whether changes in appetite are impacting sleep initiation (see below). Dose can be taken earlier in the day or add a small dose of an immediate-release psychostimulant medicine later in the day to reduce rebound ADHD symptoms.3, 76 Alternatively, consider reducing the dose, stopping the medicine or prescribing melatonin.3
Mood changes – e.g. irritability, dysphoria, that develop with psychostimulant medicines can be more challenging to address.2 Stopping or switching to a non-psychostimulant medicine is often required.
Changes in appetite or weight loss – recommend a balanced diet with consistent meals and snacks.63, 76 Potential strategies to manage appetite reduction include increasing calorie intake (ideally with input from a dietitian) or taking doses with food (instead of before meals).1, 3 Trialling a dose reduction or switching to a different medicine could also be considered but stop psychostimulant medicines if the patient is persistently struggling to maintain weight.1, 2
Increases in blood pressure or heart rate – reduce the dose if persistent tachycardia (> 120 beats per minute), clinically significant increases in blood pressure from baseline on two separate occasions or new-onset arrhythmias occur.63 Arrange an ECG as appropriate. Discussion with, or referral to, a cardiologist is recommended if these effects persist despite dose reduction.
Tics – initiation of psychostimulant medicines potentially unmasks a susceptibility to tics; this may be linked to emotions and the fluctuating nature of tics.2 Establish if the tics are more disruptive than baseline ADHD symptoms. If they are not, monitor for a further three months to determine if the tics are related to the psychostimulant medicines.3 If they persist, consider reducing the dose or switching to a non-psychostimulant medicine (after discussion with a psychiatrist), although resolution of tics may not occur with treatment cessation in all cases. N.B. Some patients who experience tics at baseline may report improvement due to reductions in anxiety and stress.2
Seizures – stop psychostimulant medicines and review medicine history and assess for other potential causes.63 Psychostimulant medicines can be reintroduced once the seizures are controlled if they have been ruled out as the cause.63
Psychosis or mania – stop psychostimulant medicines immediately and refer for urgent psychiatric assessment if the patient develops an acute psychotic or manic episode1, 76
 Red flags that require input from secondary care
Red flags that require input from secondary care
Ongoing management of patients with ADHD in primary care is the goal, however, there may be times when specialist advice or care is required. Situations where discussion with, or referral to, a psychiatrist (or other appropriate specialist, e.g. cardiologist) are recommended include:76
- Difficulty managing symptoms despite adequate medicines trials with optimised doses
- Development of, or worsening, psychiatric symptoms
- New-onset cardiovascular symptoms (that do not improve with dose reduction), or a change in cardiovascular diagnosis
- Unintentional body weight reduction of 5% or more (depending on the patient’s starting BMI)
- Strong suspicion or confirmation of misuse or diversion of ADHD medicines
Trial a different psychostimulant if treatment response is insufficient
Not all patients will respond to the first psychostimulant medicine they trial. Patients who do not experience sufficient (or any) improvement in ADHD symptoms after a six-week trial of one psychostimulant at an appropriate dose (e.g. 0.5 – 1 mg/kg), should be switched to another formulation or medicine.1 Evidence for adults is limited, but an analysis of some small studies suggests that approximately 70% of children living with ADHD respond to the first psychostimulant medicine trialled, and more than 90% respond to at least one psychostimulant medicine.77
Before switching to a different psychostimulant medicine:2, 78
- Determine that the medicine is being taken as prescribed, e.g. correct dose at the appropriate time
- Confirm that the agreed upon treatment goals are realistic and align with the expected effects of psychostimulant medicines
- Assess whether the most appropriate ADHD symptoms are being measured to evaluate treatment response
- Identify modifiable reasons that treatment goals are not being achieved and adjust the dosing schedule where possible, e.g. timing the maximum treatment effect to occur when it is most needed, changing to a longer-acting formulation so that the patient does not have to take a dose at work or suggesting weekly pill boxes to help with organisation
- Determine if other factors could be affecting treatment, e.g. psychiatric co-morbidities, family or social circumstances
- Reconsider if the ADHD diagnosis is correct
Due to differences in pharmacokinetics between formulations of methylphenidate as well as inter-person variability in treatment response clinicians are generally advised to specify the brand of methylphenidate when prescribing to avoid patients experiencing insufficient treatment response or adverse effects due to unnecessary brand changes. However, recent global supply issues have meant that switching between brands is often necessary; therefore, the advice has been updated to consider prescribing immediate-release methylphenidate generically where appropriate to make this process easier.30 Modified-release methylphenidate preparations must still be prescribed by brand.30
Methylphenidate and amfetamines are not considered dose equivalent.* 29 If the decision is made to switch psychostimulant medicines, start at the lowest dose of lisdexamfetamine or dexamfetamine and slowly increase the dose based on treatment response and adverse effects.29 Non-psychostimulant treatment options such as atomoxetine should generally be reserved until all psychostimulant medicines have been trialled at adequate doses (or for an adequate duration), unless there is a clinical indication for using them earlier, e.g. psychostimulant medicines are contraindicated or not tolerated.1
* Expert opinion is that dexamfetamine doses are typically half of methylphenidate doses providing an optimum dose target to guide titration; starting at a low dose and titrating up when switching between medicines is still strongly recommended.
For further information on bioequivalence and switchability, see: A reminder: generic medicines, bioequivalence and switchability (Medsafe)
General principles for switching psychostimulant formulations
Caution is recommended when switching patients to a different brand of medicine in any clinical situation.25 Advice for switching between psychostimulant medicine formulations in patients with ADHD is largely based on clinical experience.
Key considerations include:
- Establish a baseline for symptoms and daily functioning to guide dose titration for the new formulation (see: “Scales for monitoring treatment response”).63 Re-evaluation for substance misuse or diversion may also be appropriate.
- Check the patient’s medical history to see if they have previously trialled other brands of methylphenidate or amfetamines, and if so, how they responded. Brands (or medicines) that previously resulted in a good treatment response and were well tolerated should be the first choice when switching.
- Ideally, select another brand that is most similar to the current medicine (e.g. if prescribed the extended-release methylphenidate, Concerta, choose another extended-release brand, i.e. Methylphenidate Sandoz XR or Methylphenidate ER – Teva; see Table 2). However, be aware that some patients may not respond/experience the same effect even when they change to a similar brand; close monitoring and regular follow-up is required.
- Theoretically, all brands of methylphenidate have daily dose equivalence, i.e. 20 mg of immediate-release methylphenidate taken in divided doses is equivalent to 20 mg of sustained-release methylphenidate. However, a new brand is often started at a reduced dose, to evaluate treatment response before titrating back up. If switching from an immediate-release to a longer-acting formulation, calculate the total daily dose and delivery over a similar duration, e.g. a patient prescribed 10 mg in the morning and early afternoon could be switched to a 20 mg dose of a modified-release formulation, e.g. Rubifen SR or Ritalin LA.
- If switching from a modified-release (or prodrug) formulation to divided doses of an immediate-release formulation, warn patients that they may experience variations in symptom control because of peaks and troughs in plasma levels caused by repeated dosing
- Close monitoring is required whenever a patient is switched to a different psychostimulant medicine (brand or formulation type).24 Evaluate treatment response and adverse effects, e.g. appetite suppression, mood swings and sleep disturbance, one to two weeks after any change in formulation or dose.
- Consider dosing adjustments before switching again. Confirm that any new adverse effects are not related to the duration of action, e.g. the patient is experiencing insomnia because the duration of action is too long (or too short, i.e. the patient cannot “shut off” their brain before bed), or low mood and irritability due to a rapid reduction in plasma level (rebound effect). Adjust the dosing schedule if required.
- Trial a second alternative within the same class before switching to a different medicine if the patient does not respond/tolerate the first formulation, e.g. trial two brands of methylphenidate before switching to an amfetamine, or trial both types of amfetamine before switching to a methylphenidate formulation.
Best Practice Tip: Contact the patient’s preferred pharmacy before prescribing a different brand, formulation type or medicine to confirm what stock is currently available, and work with your local pharmacies to establish the best way of staying informed about their stock levels. Latest information on supply issues is also available on the Pharmac website.
Dispensing considerations in relation to supply shortages
Pharmacists must navigate ongoing supply issues, as well as controlled drug regulations and funding rules when dispensing psychostimulant medicines. Regulation 42 (4) of the Medicines Regulations 1984 permits brand substitution for psychostimulant medicines providing that the active ingredient, dose form and strength remain the same, and there is no clinical reason that the new brand should not be dispensed. Key considerations if the prescribed brand of psychostimulant is not available:79
 Dispensing a different brand of immediate-release methylphenidate is permitted without a new prescription as long as the total dose for each individual dose remains unchanged, e.g. substituting Rubifen 10 mg where Ritalin 10 mg is not available
Dispensing a different brand of immediate-release methylphenidate is permitted without a new prescription as long as the total dose for each individual dose remains unchanged, e.g. substituting Rubifen 10 mg where Ritalin 10 mg is not available
 Dispensing different strengths of the same formulation of psychostimulant is permitted without a new prescription as long as the total dose for each individual dose remains unchanged, e.g. dispensing two Ritalin LA 20 mg capsules per dose when Ritalin LA 40 mg capsules are prescribed but unavailable
Dispensing different strengths of the same formulation of psychostimulant is permitted without a new prescription as long as the total dose for each individual dose remains unchanged, e.g. dispensing two Ritalin LA 20 mg capsules per dose when Ritalin LA 40 mg capsules are prescribed but unavailable
 Dispensing a different brand of modified-release methylphenidate is not permitted and prescriber involvement is required for consideration of switching to a different modified-release formulation. A new prescription is required in these situations.
Dispensing a different brand of modified-release methylphenidate is not permitted and prescriber involvement is required for consideration of switching to a different modified-release formulation. A new prescription is required in these situations.
Where a permitted change is made to a prescription, pharmacists must annotate the change on the prescription, e.g. brand supplied, date of change, signature of pharmacist authorising the change.79
Regularly revisit the need for treatment
Treatment should be continued as long as the patient is experiencing benefit; this may be life-long in some cases.1 Evidence of the positive effect of long-term psychostimulant medicines on patient quality of life and adverse outcomes is beginning to emerge, however, there is currently not considered to be a strong evidence base supporting the benefit of psychostimulant medicines on core ADHD symptoms over placebo after 12 months (see: “Read the evidence: Medicines for ADHD”).3, 33, 34 There are also reports that the effects of treatment begin to “wear off” after a period for some people.1, 3 The pathophysiology underlying this tolerance is unknown but may involve downregulation of dopamine receptors.3
Regularly discuss preferences for continuing or stopping treatment with the patient (and caregiver if relevant).1 Specific recommendations on how often this should occur range from annually to every two to four years.1, 57 Base the decision on:1
- Confirmation that a clinical indication remains
- The patient’s opinion on whether they are still experiencing a therapeutic benefit. Parent/caregiver or teacher opinion may be relied upon in younger people.
- The development of any new adverse effects
- The impact of missed doses or dose reductions on the patient’s day-to-day life
As part of this review, consider a treatment holiday (see below) or a period of treatment at a reduced psychostimulant dose.1
Treatment holidays should be considered
Guidelines suggest that patients with ADHD who have been stabilised on an optimum dose of a psychostimulant medicine undergo a short treatment-free period, however, this is based on limited evidence.1, 2 A treatment holiday is useful to determine the ongoing need for treatment and guide decisions around discontinuing treatment; it has also been suggested it may reduce potential tolerance to psychostimulant medicines.80
These trials should take place during a period of “low cognitive load”, e.g. a holiday break, or on weekends.2 The optimal duration for a treatment holiday is unclear. Up to one to two weeks has been suggested but ultimately the decision on how long to stop treatment for should be made between the prescriber and patient and include options for consultation and restarting treatment, if required. In some cases, a treatment break can be trialled during normal work or study periods to evaluate the impact of ADHD symptoms on performance and provide a clearer understanding of ongoing need for treatment.
Stopping psychostimulant treatment for ADHD
Choosing to stop treatment completely can occur for several reasons, such as the patient’s opinion on treatment changing, their symptoms no longer being severe enough or experiencing adverse effects that outweigh the benefits of treatment.2 Psychostimulant medicines can generally be stopped abruptly without adverse effects. A gradual taper may be considered if the patient is stabilised on a high dose, or has previously reported discontinuation symptoms during a treatment holiday.2
Best Practice Tip: Patients may want to restart treatment in the future. Discuss factors that may prompt a discussion about restarting psychostimulant medicines, e.g. symptoms are impacting day-to-day life or there are changes in family, employment or education circumstances.
General practitioners and nurse practitioners working in primary care cannot initiate ADHD treatment in children and adolescents aged under 18 years without a written recommendation but still have a significant role in the continuation of treatment and ongoing monitoring. Many of the principles in this article apply to all patients with ADHD, however, there are some aspects of the ongoing prescribing of psychostimulant medicines where extra consideration for children and adolescents should be made.
Methylphenidate is the preferred treatment choice
Methylphenidate is the first-line treatment option for children with ADHD.32, 81 It is recommended over amfetamines in younger people due to its improved tolerability, despite being considered marginally less effective.32, 81 This aligns with Pharmac guidance during current supply issues advising to prioritise methylphenidate for children and adolescents.9
Monitoring and follow-up of children and adolescents prescribed psychostimulant medicines
Review should occur at least every six months.81 Insufficient gain in height or weight, severe ADHD symptoms, co-morbid conditions or medicines adherence or abuse/diversion concerns should prompt more frequent assessment. Ongoing monitoring and re-evaluation of the need for pharmacological treatment in children should follow a similar approach to adults (see: “Monitoring and follow-up of adults stabilised on psychostimulant medicines”).
Key differences when monitoring children in primary care compared to adults include:60, 85
ADHD symptoms. Use child-specific scales to measure therapeutic effect over time (see: “Scales for monitoring treatment response”).63
Adverse effects. Common adverse effects of psychostimulant medicines in children include headaches, abdominal pain, sleep disturbances, appetite suppression, aggression and mood changes, e.g. anxiety or irritability.81 These will often improve with time;81 consider a dose reduction if adverse effects are intolerable or persist. Some children may become subdued and avoid social interaction when taking psychostimulant medicines;81 switching to a different psychostimulant or non-psychostimulant medicine may be appropriate and should be discussed with a paediatrician or paediatric psychiatrist.
Consider prescribing melatonin (unapproved indication in children) if dose adjustment does not improve sleep disturbances in children. Expert opinion is that modified-release formulations of melatonin can be crushed for children who cannot take tablets, however, this changes the release profile to act more like immediate-release melatonin.
Measuring weight and height. Weight should be measured at three and six months after initiation of treatment and then every six months thereafter.1 Increasing calorie intake (ideally under dietitian supervision), nutritional supplements, more frequent monitoring or treatment holidays are appropriate strategies to address inadequate weight gain in children.1, 2, 82 Taking psychostimulant medicines during development may also affect growth rates and terminal adult height.81 Regular measurement of height is therefore critical to identify any deviations from expected growth trajectories.81 Discussion with, or referral to, a paediatrician is recommended if changes in growth rates are identified.81 A treatment holiday could be considered in this situation however, there is limited evidence to support this.1, 82
Heart rate and blood pressure. Psychostimulant medicines may cause small increases in heart rate and blood pressure in children and adolescents; mean elevations in heart rate (≤ 10 beats per minute) and blood pressure (≤ 5 mmHg) have been reported.81, 83 Up to 15% of children taking psychostimulant medicines may experience larger increases in these parameters.83 Compare recorded heart rate and blood pressure values with accepted normal values for children of the relevant age and sex.1 Persistently elevated values warrant discussion with, or referral to, a paediatrician.1
Risk of diversion and misuse. The potential for misuse and abuse of psychostimulant medicines changes as children get older. It may be appropriate to reassess the risk of diversion and misuse, depending on age of the patient.63
- Earlier initiation of psychostimulant medicines in children, i.e. prior to age nine years, and a longer duration of treatment may reduce the risk of substance misuse in adolescence84
Best Practice Tip: If dosing adjustments are required, suggest that caregivers make the change at the start of the weekend, i.e. Saturday morning. This allows time to monitor the child’s response before they return to school on Monday.
Clinician resources
Patient and family/whānau resources
N.B. Ask the patient what their preferred medium for receiving health information is. Conventional patient handouts and information leaflets may not be the best method to provide information to patients living with ADHD. Consider referring them to websites, podcasts or books that may be helpful.
Table 2. Overview of funded psychostimulant medicines available for managing ADHD in adults and children aged six years and over as of 1st February, 2026.1, 2, 23, 25, 30, 35–43 N.B. The data in this table are compiled from the New Zealand Formulary (and New Zealand Formulary for Children), the ADHD Prescribing Guide for Australian Healthcare Professionals, New Zealand data sheets and expert opinion, and should only be used as guide.
|
| Medicine |
Methylphenidate |
Dexamfetamine
(unapproved indication in adults) |
Lisdexamfetamine |
| Special Authority application form: |
SA2590
(Rubifen LA will be included on this form if funded) |
SA2591 |
SA2587 |
SA2588 |
| Brand: |
Ritalin |
Rubifen |
Rubifen SR |
Methylphenidate ER - Teva |
Methylphenidate Sandoz XR (provisionally approved until September, 2027) |
Rubifen LA
(proposed to be funded from 1st July, 2026) |
Ritalin LA |
Concerta |
Dexamfetamine (Noumed) |
Vyvanse |
| Medicine type: |
Innovator |
Generic (of Ritalin) |
Generic (of Ritalin SR) |
Generic (of Concerta) |
Generic (of Concerta) |
Generic (of Ritalin LA) |
Innovator |
Innovator |
Generic |
Innovator |
| Formulation and strengths available: |
Tablet
10 mg |
Tablet
5 mg
10 mg
20 mg |
Modified-release tablet
20 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Modified-release capsule
10 mg
20 mg
30 mg
40 mg
60 mg |
Modified-release capsule*
10 mg
20 mg
30 mg
40 mg |
Modified-release tablet
18 mg
27 mg
36 mg
54 mg |
Tablet
5 mg |
Capsule†
30 mg
50 mg
70 mg |
| Onset of action: |
20 minutes – 1 hour |
20 minutes – 1 hour |
1 – 2 hours |
1 – 2 hours |
1 – 2 hours |
1 – 2 hours‡ |
1 – 2 hours |
1 – 2 hours |
20 minutes – 1 hour |
60 – 90 minutes |
| Duration of action: |
3 – 5 hours |
3 – 5 hours |
Up to 8 hours |
Up to 12 hours |
Up to 12 hours |
6 – 8 hours |
6 – 8 hours |
Up to 12 hours |
4 – 6 hours |
8 – 14 hours |
| Contraindications (for the full list of contraindications, see the NZF or New Zealand data sheets): |
- Acute severe depression, uncontrolled bipolar disorder, suicidal ideation or psychosis – patient should be stabilised first (involving specialist care)
- Anorexia nervosa
- Uncontrolled substance dependence or alcohol misuse (including in family and caregivers), however, expert opinion is that treatment could be carefully considered as part of a complete treatment plan if this is well-controlled or in remission (under specialist supervision)
- Hyperthyroidism
- Pre-existing cardiovascular disease, e.g. advanced arteriosclerosis, heart failure, myocardial infarction, arterial occlusive disease, cardiomyopathy, moderate to severe hypertension, arrhythmias, structural cardiac abnormalities
- Cerebrovascular disorders including aneurysm, vasculitis, vascular abnormalities, stroke — due to effects on heart rate and blood pressure
- Acute angle-closure glaucoma
- Hypersensitivity to the active ingredient
Note: Modified-release tablets should not be prescribed to patients with dysphagia or restricted size of gastro-intestinal lumen, e.g. gastric bypass |
|
|
| Cautions (for the full list of cautions, see the NZF or New Zealand data sheets): |
Psychostimulant medicines may precipitate or worsen:
- Anxiety or agitation
- Epilepsy
- Mild hypertension (or other cardiovascular conditions that may be compromised by increases in blood pressure or heart rate)
- Psychiatric disorders, e.g. bipolar disorder, or aggressive behaviour
- Tics – an association between psychostimulant medicine and development of tics was previously established but evidence now suggests this effect is no greater than placebo (or tic-specific treatments).44 The development of tics may be related to the emotions surrounding initiating treatment and the fluctuating nature of tics (caution is also recommended if the patient has a family history of Tourette syndrome).
Caution is also recommended when prescribing psychostimulants to patients with:
- Past history of disordered eating or difficulty maintaining weight
- Past history of substance or alcohol dependence or misuse (including in family and caregivers)
|
Key interactions
(for the full list of interactions, see the NZF or New Zealand data sheets): |
- Monoamine oxidase inhibitors, e.g. tranylcypromine, moclobemide
- Concurrent use can lead to hypertensive crisis – do not use within 14 days
- Tricyclic antidepressants
- Concurrent use may increase plasma concentrations of tricyclic antidepressants and increase the risk of adverse effects, e.g. cardiovascular effects
- Selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs)
- Concurrent use may increase the risk of serotonin syndrome
- Medicines that influence blood pressure
- Blood pressure control may be affected in patients taking antihypertensives, e.g. amlodipine, losartan, quinapril
- Increased risk of hypertension with concurrent use of pseudoephedrine, phentermine
|
- Opioids, e.g. codeine, tramadol
- May increase analgesic effect
- Antipsychotics, e.g. chlorpromazine
- May reduce the effect of amfetamines
|
Key adverse effects
(for the full list of adverse effects, see the NZF or New Zealand data sheets): |
Common:
- Insomnia
- Nausea and stomach discomfort, reduced appetite
- Sweating
- Hypertension, tachycardia and palpitations
- Anxiety, agitation, depressed mood, dysphoria, headache, irritability, tics – establishing baseline symptoms before initiating treatment is critical to distinguish adverse effects of psychostimulant treatment
Rare:
- Hepatic dysfunction
- Angina, myocardial infarction, supraventricular tachycardia
- Psychosis
- Cerebrovascular disorders, e.g. vasculitis, haemorrhage, cerebral arteritis, and vascular occlusion
- Tourette syndrome (in predisposed individuals)
- Seizures
- Angle-closure glaucoma
|
| Starting dose: |
5 mg, two to three times daily |
5 mg, two to three times daily |
20 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
20 mg, once daily in the morning |
20 mg, once daily in the morning |
18 – 36 mg, once daily in the morning |
2.5 mg, two to three times daily |
20 – 30 mg, once daily in the morning |
| Suggested titration based on response: |
Increase dose by 5 mg, weekly |
Increase dose by 5 mg, weekly |
Increase dose by 20 mg, weekly |
Increase dose by 18 mg, weekly |
Increase dose by 18 mg, weekly |
Increase dose by 20 mg, weekly (in adults) |
Increase dose by 20 mg, weekly (in adults) |
Increase dose by 18 mg, weekly |
Increase dose by 5 mg, weekly |
Increase dose by 10 – 20 mg, at a minimum of weekly intervals |
| Maximum dose: Discussion with, or referral to, a psychiatrist recommended if higher doses are being considered |
60 mg, divided over two to three doses, daily |
60 mg, divided over two to three doses, daily |
60 mg, once, daily |
72 mg, once daily |
72 mg, once daily |
80 mg, once daily |
80 mg, once daily |
72 mg, once daily |
20 mg, divided over two to three doses, daily, however, doses of 30 mg per day are commonly used
(up to 40 mg/day required in some children) |
70 mg, daily |
| Excipients with known effects or derived from animal sources: |
Gelatine, gluten, lactose |
None |
Lactose |
Lactose |
Lactose |
Gelatine |
Gelatine |
Lactose |
Lactose |
Gelatine |
| Comments: |
Can be taken with or without food |
Can be taken with or without food |
Should be taken with food – onset of action is faster when taken with a high-fat meal
Fewer fluctuations in plasma concentrations compared to multiple doses of immediate release methylphenidate |
Can be taken with or without food |
Can be taken with or without food |
Can be taken with or without food
Capsules can be opened, and beads can be sprinkled on cold food if required for administration |
Can be taken with or without food
Capsules can be opened, and beads can be sprinkled on cold food if required for administration |
Can be taken with or without food |
Can be taken with or without food |
Capsules can be opened and contents dissolved in water or juice, or mixed through food if required for administration |
* 60 mg Ritalin LA capsules are also available but not funded
† 20 mg, 40 mg and 60 mg lisdexamfetamine capsules are also available but not funded
‡ Rubifen LA is a generic of Ritalin LA and assumed to have the same onset and duration of action as the innovator
Best Practice Tip: Caution is recommended when switching between brands of methylphenidate and close monitoring and more frequent follow-up is advised. Generic medicines have demonstrated comparable bioavailability to innovator medicines and are considered bioequivalent by Medsafe. However, some patients may experience differences in effect when switching between innovator and generic medicines due to the medicine’s therapeutic index, variation in pharmacokinetics or patient populations, or patient factors, e.g. the nocebo effect. Clinicians with experience in prescribing these medicines have observed that differences in effect can occur when switching between formulations of methylphenidate, which is also noted in the New Zealand Formulary.





