Serum sodium imbalance
Key concepts:
- Further laboratory investigations may be appropriate if the clinical assessment and patient history do not reveal
the cause of the sodium imbalance
- Serum sodium imbalances are more prevalent in older people and hyponatraemia is more commonly seen in general practice
than hypernatraemia
- The cause of the sodium imbalance is usually clinically apparent, e.g. fluid depletion or overload
- Urgent referral to secondary care is recommended for patients with serum sodium < 120 mmol/L or > 150 mmol/L,
rapidly decreasing or increasing levels, if neurological symptoms are present or if the patient is systemically unwell
The normal reference range for serum sodium for adults is 135 – 145 mmol/L
Understanding sodium imbalance
Sodium is essential in the human body. It has a vital role in maintaining the concentration and volume of the extracellular
fluid and accounts for most of the osmotic activity of plasma. Serum sodium levels are maintained by feedback loops involving
the kidneys, adrenal glands and hypothalamus.
When serum sodium is low (usually because total body water is high), antidiuretic hormone (ADH) is suppressed and a
dilute urine is excreted.1 In addition, the kidney produces renin, which stimulates aldosterone production,
which decreases the excretion of sodium in the urine, therefore increasing sodium levels in the body.
When serum sodium is high (usually because total body water is low), ADH is released, causing the kidneys to conserve
water and therefore a concentrated urine is excreted.1 In addition, atrial natriuretic peptide (ANP) is secreted
by the heart (in response to high blood pressure caused by increased sodium levels) and promotes loss of sodium by the
kidney (by inhibiting renin and therefore aldosterone secretion).
In people with normal renal function and adequate aldosterone production, fluid and electrolyte balance is able to be
maintained in the body through these compensation processes. Sodium imbalances can indicate the presence of an underlying
medical condition or the affect of a medicine.
Elderly people are more susceptible to sodium imbalances due to age-related decrease or decline in:1
- Total body water
- Thirst mechanism
- Maximal urinary concentrating ability
- Ability to excrete water load
- Renal function
Elderly people also commonly have multiple co-morbidities that can affect sodium levels and renal function. In addition,
use of medicines that affect electrolyte excretion or retention, e.g. diuretics, also commonly causes sodium imbalance.1
Low serum sodium levels – Hyponatraemia
Hyponatraemia is defined as a serum sodium concentration < 135 mmol/L
Severe hyponatraemia is defined as a serum sodium concentration ≤ 120 mmol/L
Hyponatraemia is the most common electrolyte disorder and is often an incidental finding on routine blood tests.
Hyponatraemia describes a serum sodium concentration which is lower than normal (< 135 mmol/L). It is most commonly
a result of excess water diluting the serum sodium levels in the body (e.g. as seen in congestive heart failure), but
hyponatraemia can also exist with normal or decreased water levels.
Mild, asymptomatic hyponatraemia does not usually require corrective measures except for treatment of the underlying
factors. Correction of hyponatraemia, when required, is usually done in secondary care. Treatment must be gradual to avoid
the risk of both fluid overload and cerebral demyelination, which can be fatal.
Signs and symptoms of hyponatraemia
Signs and symptoms of hyponatraemia are generally related to the underlying cause, whether or not it is associated with
fluid loss or dehydration, the degree of hyponatraemia and the rate at which it develops.
The signs and symptoms of mild hyponatraemia are usually non-specific, e.g. nausea and lethargy. People with mild, long-term
hyponatraemia are often asymptomatic.2 Severe (serum sodium < 120 mmol/L) or rapid-onset hyponatraemia can
be associated with disorientation, agitation, unsteadiness, seizures, coma and death, due to cerebral oedema.1,2
When hyponatraemia is associated with decreased extracellular fluid then signs and symptoms can include dizziness, postural
hypotension and dry mucus membranes.
Medical disorders that can cause hyponatraemia
Conditions that can cause hyponatraemia include: gastroenteritis, pneumonia, anorexia nervosa, renal disease, hypothyroidism,
Addison’s disease, congestive heart failure, liver disease, myeloma, small cell lung cancer, lymphoma, stroke,
tumour, meningitis.4,5
Assessing a patient with hyponatraemia
Assess the level of severity
Refer the patient to secondary care for treatment if sodium < 120 mmol/L.
Assess the trend
Check for previously low serum sodium measurements or repeat the test if time permits. A rapid decrease in sodium
warrants referral to secondary care even if the actual degree of hyponatraemia is only moderate.3
N.B. changes of up to 5 mmol/L in two sequential individual results can reflect non-significant variation in sodium
levels.3
Assess clinical status
Assess for signs and symptoms indicative of cerebral oedema, e.g. increasing confusion, decreasing consciousness,
seizures. If present, urgent transfer to hospital is indicated.3
Assess if there is any acute illness, e.g. pneumonia, gastroenteritis.
Assess hydration status. Check for dehydration, postural changes in blood pressure,
jugular venous pressure, peripheral oedema and ascites. Ask about fluid intake/loss and increased/decreased thirst.
Consider known conditions that may have caused the hyponatraemia, e.g. congestive heart failure, renal or liver disease.
Assess the medication history
Look for medicines usually implicated in hyponatraemia, e.g. diuretics, selective serotonin reuptake inhibitors
(SSRIs).
Determining the cause of hyponatraemia
After assessing the patient, the cause of the hyponatraemia is usually evident.
Patient is hypervolaemic (i.e. fluid overload): consider possible causes such as liver cirrhosis, congestive
heart failure, renal failure and nephrotic syndrome.
Patient is euvolaemic (i.e. normal fluid status): consider possible causes such as medicines, water
intoxication, renal failure, hypothyroidism, glucocorticoid deficiency, syndrome of inappropriate anti-diuretic hormone
secretion (SIADH).1
If the patient is hypovolaemic (i.e. fluid depletion): consider possible causes such as vomiting, diarrhoea,
renal disease, diuretics, pancreatitis, burns, disorders of CNS causing salt wasting.
Additional laboratory tests may be useful if no obvious cause is found
If no obvious cause for the hyponatraemia can be found, additional blood and urine tests may be useful. Discussion with
a renal physician is recommended.
Normal results of lipids, glucose, protein and albumin can help exclude rare causes
of low sodium levels, which can occur with marked hypertriglyceridaemia, hyperglycaemia and hyperproteinaemia. A TSH may
reveal hypothyroidism which can also be a rare cause of hyponatraemia. Abnormal LFTs may indicate cirrhosis.
Urinary sodium concentrations may be useful to help determine if the loss is renal or extra-renal.
Medicines that can cause hyponatraemia
Medicine-induced hyponatraemia usually develops within the first few weeks of starting treatment. Once the medicine
is stopped, the hyponatraemia will usually resolve within two weeks (levels can then be rechecked).6 In many
cases, a combination of medicines is responsible for the hyponatraemia rather than just one implicated agent.
Diuretics cause hyponatraemia in approximately 20% of people who take them,7 although severe
hyponatraemia is nearly always seen with thiazide rather than loop diuretics.8
Selective serotonin reuptake inhibitors (SSRIs) cause hyponatraemia in up to one-third of people who
take them. Risk factors include older age, female gender, concomitant use of diuretics, low body weight and lower baseline
serum sodium concentration. Serum sodium level should be checked before and several weeks after starting a SSRI in older
patients and in those taking other medicines associated with hyponatraemia.9
Antipsychotics are associated with polydipsia (increased thirst), which in turn can cause hyponatraemia.
A history of polydipsia has been found in 67% of people taking antipsychotic medicines.10
Non-steroidal anti-inflammatory drugs (NSAIDs) can cause water retention by increasing water permeability
across the renal collecting ducts.11
Other medicines associated with hyponatraemia include: carbamazepine, tricyclic antidepressants, ACE
inhibitors, angiotensin II receptor blockers (ARBs), proton-pump inhibitors, sulphonylureas, dopamine agonists, opiates,
amiodarone, some chemotherapy medicines, e.g. vincristine, vinblastine, and high dose cyclophosphamide.11
High serum sodium levels – Hypernatraemia
Hypernatraemia is defined as a serum sodium level of > 145 mmol/L
Severe hypernatraemia is defined as a serum sodium level of > 155 mmol/L
Hypernatraemia is much less commonly encountered in general practice than hyponatraemia but when it does occur it is
associated with a high mortality rate.
Hypernatraemia describes a serum sodium concentration which is higher than normal (>145 mmol/L). It is characterised
by a deficit of water in relation to sodium in the body, which can result from either a net water loss (which would usually
be corrected by increased fluid intake via the thirst mechanism) or less commonly, a hypertonic sodium gain. In most cases
the cause of the hypernatraemia will be apparent from the clinical setting. Common causes include kidney disease, inadequate
water intake and loss of water through vomiting or diarrhoea.
People at highest risk of hypernatraemia include:
- Infants and elderly people who cannot maintain adequate fluid intake without assistance
- People with impaired mental status who are unable to ask for water
- People with uncontrolled diabetes
- People with an impaired thirst mechanism
- Hospitalised patients receiving hypertonic infusions, tube feedings, osmotic diuretics, lactulose or mechanical ventilation
Signs and symptoms of hypernatraemia
The signs and symptoms of hypernatraemia are primarily neurological and can include lethargy, weakness and irritability.
With more severe hypernatraemia or a rapid rise in sodium level, this can progress to twitching, seizures, coma and death.1
Clinical evidence of dehydration may be evident, e.g. tachycardia, low blood pressure and decreased urine output. Symptoms
in older people may be non-specific.1
Assessing a patient with hypernatraemia
Assess the severity
Refer the patient to secondary care for treatment if the serum sodium is ≥ 155 mmol/L.3
Assess the trend
Check for previous results or if time permits, repeat levels. Refer to secondary care if levels are rapidly increasing.
Assess clinical status
If neurological symptoms are present, if the patient is systemically unwell or if oral rehydration is not possible,
refer to secondary care for treatment.3
Assess if there is any acute illness, e.g. gastroenteritis.
Assess hydration status (Above).
N.B. Most patients with mild hypernatraemia caused by fluid loss or decreased fluid intake can be managed in primary
care with oral rehydration (with a balanced electrolyte solution). It is important that rehydration is performed slowly.
Excessively rapid correction or overcorrection of hypernatraemia increases the risk of iatrogenic cerebral oedema.
Assess the medication history
Look for medicines that may be implicated in hypernatraemia, e.g. loop diuretics, lithium.3
Determining the causes of hypernatraemia
The cause of hypernatraemia is usually derived from the clinical assessment and the patient’s history.
Net water loss can be due to:12,13
- Unreplaced insensible loss (dermal and respiratory)
- Inadequate fluid intake/impaired thirst – typically in elderly people
- Neurogenic diabetes insipidus – post-trauma, idiopathic, caused by tumours, sarcoidosis
- Nephrogenic diabetes insipidus – congenital or acquired (e.g. renal disease), medicines such as lithium, amphotericin
B
Hypotonic fluid loss can be due to:12,13
- Renal causes, e.g. osmotic diuresis in uncontrolled diabetes
- Medicines, e.g. loop diuretics, mannitol, urea, corticosteroids (increase production of urea), high protein supplements
- Gastrointestinal losses, e.g. diarrhoea, vomiting, fistulae, use of osmotic laxatives (lactulose, sorbitol)
- Cutaneous loss, e.g. burns, excessive sweating
Hypertonic fluid gain can be due to:12,13
- Ingestion of salt, salt water, sodium rich enemas
- IV hypertonic infusions, e.g. sodium bicarbonate, sodium chloride
Additional investigations
Additional investigations are indicated by the clinical situation and may include: serum potassium, urea, creatinine,
calcium and glucose.3
Osmolality testing
Serum and urine osmolality tests are frequently mentioned in the literature as part of the investigation for either
hyponatraemia or hypernatraemia. However, these tests would rarely be requested in the general practice setting.
Urine osmolality is used to detect the ratio of water and solutes in the urine (using a random urine specimen). Urine
osmolality is controlled by ADH and it varies over a wide range to reduce the effect of fluid intake on serum osmolality,
which is tightly controlled.14 A high urine osmolality (> 600 mOsm/kg) can indicate fluid losses (e.g. gastrointestinal,
diuretics) and a low urine osmolality (< 300 mOsm/kg) can indicate diabetes insipidus or water diuresis.1,14

Serum osmolality measures the ratio of water and solutes in the serum and is used to determine hydration status. Paired
serum and urine osmolality samples can be used to investigate causes of hyponatraemia. A decreased serum osmolality (i.e. < 280
mOsmol/kg) indicates true hyponatraemia. If the urine osmolality is > 100 mOsmol/kg, this can suggest SIADH.2,14
Urine sodium concentration indicates whether losses are renal or extra-renal and can be used to further interpret results.2
Serum potassium imbalance
Key concepts:
- Serum potassium imbalance is more prevalent in older people and people with co-morbidities, e.g. renal impairment,
congestive heart failure
- Both hypokalaemia and hyperkalaemia can cause cardiac arrhythmias which may be life threatening
- Urgent referral to secondary care is recommended for patients with serum potassium ≤ 2.5 mmol/L or ≥ 7 mmol/L, rapidly
decreasing or increasing levels, neuromuscular symptoms or ECG changes, or if the patient is systemically unwell
- The cause of the potassium imbalance is usually clinically apparent, e.g. vomiting and diarrhoea, secondary to medicines
or renal impairment.
- Further laboratory investigations may be appropriate if the clinical assessment and patient history do not reveal
the cause of the potassium imbalance
The normal reference range for serum potassium is 3.5 – 5.3 mmol/L
Understanding potassium imbalance
Potassium is required for the normal functioning of all excitable cells, particularly those within the heart and all
skeletal and intestinal muscles. It is necessary for brain function (regulation of neuromuscular excitability), cardiac
function (contractility and rhythm) and the maintenance of fluid and electrolyte balance.
Almost all (98%) of the body’s potassium is in intracellular fluid (predominately in muscle, liver and erythrocytes)
with the remainder circulating in the serum. This large difference in concentration between intra- and extracellular fluid
is maintained by enzymes (Na-K-ATPase) that actively pump potassium into the cell and sodium out, to maintain a serum
potassium concentration between 3.5 and 5.3 mmol/L. Any alteration in the distribution of intracellular and extracellular
potassium may, therefore, lead to either hypo- or hyperkalaemia.
The kidneys have a key role in regulating potassium balance with the proximal tubules reabsorbing nearly all of the
filtered potassium. Under the influence of aldosterone, additional potassium is secreted into the distal tubules and collecting
ducts in exchange for sodium. In a healthy adult, almost the entire daily intake of potassium is excreted with approximately
90% via the kidneys and the remaining 10% in the stool. Therefore potassium balance is largely maintained by the regulation
of excretion of potassium in the urine.15,16
Both hypokalaemia and hyperkalaemia are less common in healthy adults with normal renal function, however, in older
people, a reduction in renal function and changes in the normal homeostatic mechanisms that maintain potassium balance,
make this group more susceptible to potassium imbalance.
Low potassium levels – Hypokalaemia
Hypokalaemia is defined as a serum potassium concentration of < 3.5 mmol/L
Severe hypokalaemia is defined as a serum potassium concentration of ≤ 2.5 mmol/L
Mild hypokalaemia is often well tolerated in otherwise healthy people, however, in people with co-morbidities, particularly
those with hypertension, underlying heart disease or cirrhosis, it is associated with an increased incidence of life-threatening
cardiac arrhythmias, sudden death, and rarely hepatic coma (in people with cirrhosis).17,18
Signs and symptoms of hypokalaemia
The majority of patients with mild hypokalaemia (3.0–3.5 mmol/L) are asymptomatic and initial symptoms, when they occur,
may be non-specific such as weakness or fatigue.17 Signs and symptoms become more apparent as the potassium
level drops below 3.0 mmol/L, however, in patients where the level has decreased rapidly, or the patient is at risk of
arrhythmia, even a mild decrease in potassium may result in significant clinical problems.19
Signs and symptoms of hypokalaemia include:16,17,19
- Cardiac: hypotension, bradycardia or tachycardia, premature atrial or ventricular beats, ventricular arrhythmias,
cardiac arrest
- Muscular: decreased muscle strength, fasciculations, tetany, decreased tendon reflexes
- Gastrointestinal: constipation, signs of ileus (abdominal distension, anorexia, nausea and vomiting)
- Respiratory: hypoventilation, respiratory distress (due to effects on the respiratory muscles)
- CNS: lethargy, paralysis, paraesthesias, mental status change such as confusion, apathy and memory loss
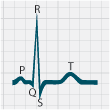
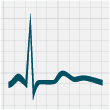
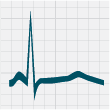
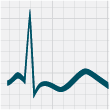
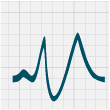
Characteristic ECG changes may occur with hypokalaemia
Hypokalaemia alters the electrical activity of cardiac muscle cells increasing membrane excitability which may cause
bradycardia, tachycardia, fibrillation, premature beats or heart block. It is recommended that an electrocardiograph
(ECG) be performed in patients with serum potassium < 3.0 mmol/L.19 ECG changes characteristically seen
in patients with hypokalaemia include: ST-segment depression, T-wave flattening, prolonged QT interval, T-wave inversion
and the presence of U waves (Figure 1).18
 |
 |
 |
 |
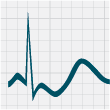 |
Normal ECG |
2.8 |
2.5 |
2.0 |
1.7 |
|
Decreasing serum potassium (mmol/L) |
Figure 1: ECG changes associated with hypokalaemia20
Assessing a patient with hypokalaemia
Assess the level of severity
Refer the patient to secondary care for treatment if potassium < 2.5 mmol/L.
Assess the trend
Check for previously low serum potassium measurements or repeat the test if time permits. A rapid decrease in
potassium warrants referral to secondary care even if the actual degree of hypokalaemia is only moderate.
Assess clinical status
Assess for signs and symptoms. If cardiac or significant CNS symptoms are present, urgent transfer to hospital
is indicated.
In moderate hypokalaemia (2.5 – 3.0 mmol/L), the need for referral will depend upon individual patient circumstances.
A suggested approach is to check for the presence of symptoms, consider an ECG and arrange a repeat blood test (same day/next
day).19
Assess if there is any acute illness, e.g. acute renal disease, gastroenteritis, diabetic ketoacidosis.
Consider known conditions that may have caused the hypokalaemia, e.g. acute or chronic renal failure, Cushing’s
syndrome.
Assess the medication history
Look for medicines usually implicated in hypokalaemia, e.g. diuretics, corticosteroids (see below).
Determining the cause of hypokalaemia
After assessing the patient, the cause of the hypokalaemia is usually evident. Diuretics are the most common single
cause of hypokalaemia.
Consider if the patient has hypokalaemia due to:16,21
Increased urinary excretion, e.g. with medicines such as diuretics or corticosteroids, diabetic ketoacidosis,
Cushing’s syndrome or disease, hyperaldosteronism.
Increased losses from other sites, e.g. primarily from the gastrointestinal tract (vomiting, diarrhoea
or intestinal fistula discharge) and also from the skin with excessive sweating.
Increased movement of potassium into the intracellular fluid, e.g. alkalosis, burns or other trauma,
medicines, e.g. high dose insulin.
Decreased intake of potassium, reduced dietary intake of potassium is a rare cause of hypokalaemia,
but may be an important factor in patients taking diuretics, e.g. an elderly patient on a “tea and toast” diet, or in
a person aiming to achieve rapid weight loss on a diet of low calorie liquid protein drinks.
Additional laboratory tests may be useful if no obvious cause is found
If no obvious cause for the hypokalaemia can be found, additional blood and urine tests may be useful. Discussion
with a renal physician is recommended.
A 24 hour collection or random urine can be used to measure urinary potassium excretion, however, these
tests are not usually ordered in general practice.23
Serum magnesium concentration could be checked as hypokalaemia is often accompanied (and made worse)
by hypomagnesaemia, e.g. in hypokalaemia due to diuretic treatment or gastrointestinal losses.19,22
Serum bicarbonate levels may help determine if an acid-base disorder is present, e.g. metabolic alkalosis.
Medicines that can cause hypokalaemia16,21,22
Diuretics – both loop and thiazide diuretics, particularly high dose, commonly cause hypokalaemia
(usually not severe). Hypokalaemia in patients taking diuretic medicines is a result of increased renal losses of potassium.
Diuretic induced hypokalaemia usually occurs within the first two weeks of treatment.
Insulin – high dose insulin (e.g. for the treatment of non-ketotic hyperglycaemia) increases intracellular
potassium and therefore decreases serum potassium
Atypical antipsychotics such as risperidone, quetapine potentiate an increase in intracellular potassium
Sympathomimetic drugs such as beta-adrenergic agonists and theophylline cause an increase in intracellular
potassium and a corresponding decrease in serum potassium
Corticosteroids – increase the renal excretion of potassium
Excessive use of laxatives – promotes increased gastrointestinal loss of potassium
A medicine that is implicated in causing hypokalaemia can be stopped if appropriate, and the potassium rechecked again
in one to two weeks. If the medicine has been stopped but the potassium level remains low, look for another underlying
cause.
High potassium levels – hyperkalaemia
Hyperkalaemia is defined as a serum potassium level > 5.3 mmol/L
Severe hyperkalaemia is defined as a serum potassium level ≥ 7.0 mmol/L or ≥ 5.4 mmol/L with
any ECG changes or symptoms
A raised serum potassium level is most commonly caused as an adverse effect of a medicine or secondary to a disease
process. Hyperkalaemia is most commonly seen in hospitalised patients,24 therefore it is less likely that patients
will present with hyperkalaemia in primary care.
Signs and symptoms of hyperkalaemia
Hyperkalaemia is often asymptomatic. When symptoms are present, they are usually non-specific such as nausea and vomiting
or less frequently muscle pain and weakness, paresthesias or flaccid paralysis. Patients may also complain of palpitations
and moderate to severe hyperkalaemia can result in cardiac disturbances and fatal arrhythmias.25
Characteristic ECG with hyperkalaemia
An ECG is recommended for patients with serum potassium levels > 6.0 mmol/L. ECG changes are not usually seen below
this level in hyperkalaemia.19
ECG changes are not always seen, even if hyperkalaemia is severe. One study showed that only 46% of patients with potassium
levels > 6.0 mmol/L had ECG changes, and only 55% of patients with potassium levels > 6.8 mmol/L had changes consistent
with hyperkalaemia.27
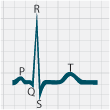
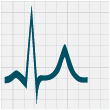
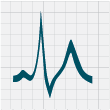
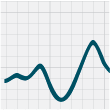
ECG changes that can be seen with hyperkalaemia include: progressive abnormalities including peaked T waves, flattening
or absence of P waves, widening of QRS complexes and sine waves (which can indicate that arrest is imminent) (Figure
2).
 |
 |
 |
 |
 |
Normal ECG |
6.5 |
7.0 |
8.0 |
9.0 |
|
Increasing serum potassium (mmol/L) |
Figure 2: ECG changes associated with hyperkalaemia20
Assessing a patient with hyperkalaemia
Assess the level of severity
Urgent referral to secondary care is recommended for patients with serum potassium ≥ 7.0 mmol/L or potassium
≥ 5.5mmol/L with any ECG changes or symptoms.26
Assess the trend
Pseudohyperkalaemia (See below) is a common reason for an isolated raised potassium level. For any
level of potassium > 6.0 mmol/L, especially if unexpected, contact the laboratory to discuss potential reasons such as
haemolysis that may explain the raised level.
Check for previously high serum potassium measurements or repeat the test if time permits. A rapid increase in potassium
warrants referral to secondary care even if the actual degree of hyperkalaemia is only moderate. Conditions that are associated
with a rapid rise in potassium (e.g. acute renal failure, rhabdomyolysis) and hypoxia of any cause are more strongly associated
with the development of cardiac conduction disturbances. Patients with potassium levels rising over six to 12 hours by > 0.5
mmol/L are considered high risk.26
Assess clinical status
Assess for signs and symptoms indicative of neuromuscular dysfunction, e.g. flaccid paralysis, paresthesias,
or impaired cardiac function, e.g. palpitations, arrhythmias. If present, urgent transfer to hospital is indicated.
It is recommended that an ECG is performed for patients with serum potassium levels > 6.0 mmol/L.19
Consider known conditions that may have caused the hyperkalaemia, e.g. acute or chronic renal failure (See
below).
Ask about any excessive dietary intake of high-potassium containing foods, e.g. dried fruits, nuts, avocado, banana,
bran cereal.
Assess the medication history
Look for medicines usually implicated in hyperkalaemia, e.g. ACE inhibitors, ARBs, spironolactone (See
below).
Determining the cause of hyperkalaemia
If urgent referral is not required, the cause of the hyperkalaemia can be investigated in primary care.
The cause will often be multifactorial, however, most cases of hyperkalaemia are associated with medicines that inhibit
the renin-angiotensin system or interfere with renal function, especially in the setting of pre-existing renal impairment.
Excessive dietary intake of potassium through food is an uncommon cause of hyperkalaemia, unless renal impairment is
present, which would decrease excretion.24
Causes of hyperkalaemia include:24,26
Renal causes
- Acute or chronic renal failure
- Mineralocorticoid deficiency (hypoaldosteronism states)
- Medicines that affect potassium excretion (amiloride, spironolactone)
- Medicines that inhibit the renin-angiotensin system (ACE inhibitors, ARBs, NSAIDs, heparin)
Transcellular shift (intracellular to extracellular compartment)
- Acidosis (including diabetic ketoacidosis)
- Medicines (digoxin poisoning, suxamethonium, beta-blockade)
Increased circulating potassium
- Exogenous (potassium supplementation)
- Endogenous (tumour lysis syndrome, rhabdomyolysis, trauma, burns)
A high potassium result can also be caused by sampling or analysis error, termed pseudohyperkalaemia.
This can result from:
- Prolonged tourniquet time or repeated fist clenching
- Test tube haemolysis
- Sample contamination with potassium EDTA (anticoagulant)
- Delayed analysis (prolonged storage of blood)
- Marked leucocytosis and thrombocytosis (measure plasma not serum concentration in these disease states)
- Sample taken from a vein infused with IV fluids containing potassium (hospital setting)
Medicine induced hyperkalaemia
Medicines are thought to contribute to the development of hyperkalaemia in the majority of cases. In one study of 242
patients admitted to secondary care with hyperkalaemia, 63% were taking medicines that affect potassium balance.27
Many cases of hyperkalaemia are related to patients with pre-existing or new renal failure who are prescribed ACE inhibitors
or ARBs in conjunction with spironolactone. These medicines increase the risk of hyperkalaemia in patients with impaired
renal function because they impair aldosterone secretion and reduce renal perfusion (and therefore the glomerular filtration
rate decreases), both of which decrease excretion of potassium by the kidneys. Approximately 10% of patients develop hyperkalaemia
within one year of starting treatment with ACE inhibitors or ARBs.27,28
Other cases of hyperkalaemia are related to potassium supplementation and prescription of diuretics or other medicines
with potassium-sparing properties.26 NSAIDs inhibit renin secretion (leading to hypoaldosteronism and reduced
potassium excretion) and can impair renal function, therefore they should be prescribed with extreme caution in people
with diabetes or renal insufficiency, particularly if they are concurrently using ACE inhibitors or ARBs.29
When prescribing medicines that may cause hyperkalaemia (Table 2) for at risk patients, start with low doses and monitor
more closely. If hyperkalaemia occurs, consider the medicine as the cause and stop treatment if possible and retest potassium
levels.
Table 2: Medicines that can cause hyperkalaemia (adpated from Nyirenda, 2009)
24
Medicines that inhibit activity of epithelial sodium channel
- Potassium sparing diuretics, e.g. amiloride
- Trimethoprim
Medicines that alter transmembrane potassium movement
- β blockers
- Digoxin
- Hyperosmolar solutions, e.g. mannitol, glucose
Potassium containing medicines
- Potassium supplements
- Salt substitutes
- Herbal medicines, e.g. alfalfa, dandelion, horsetail, milkweed, and nettle
- Stored red blood cells (haemolysis releases potassium)
Medicines that reduce aldosterone secretion
- ACE inhibitors
- Angiotensin II receptor blockers (ARBs)
- NSAIDs
- Heparin
- Antifungals, e.g. ketoconazole, fluconazole, itraconazole
- Cyclosporin
- Tacrolimus
Medicines that block aldosterone binding to mineralocorticoid receptor
- Spironolactone
- Drospirenone
Acknowledgement
Thank you to Dr Cam Kyle, Clinical Director of Biochemistry, Diagnostic Medlab, Auckland and Dr
Sisira Jayathissa, General Physician and Geriatrician, Clinical Head of Internal Medicine, Hutt Valley DHB,
Wellington for expert guidance in developing this article.