In this article 
View / Download
pdf version of this article
Update: Oxycodone: how did we get here and how do we fix it? (BPJ62)
| Key concepts: |
- Oxycodone is a strong opioid and is a second line option (after morphine) for use at step three on the WHO analgesic
ladder
- Morphine remains the first-line strong opioid and oxycodone should be reserved for specific situations
- Oxycodone can be considered if morphine is poorly tolerated
- Oxycodone is not a substitute for codeine at step two on the analgesic ladder. If the response to codeine is unsatisfactory,
morphine should be considered (i.e. step three)
- Oxycodone has a number of potentially significant drug interactions that do not occur with morphine
- Oxycodone is not completely safe in renal impairment
|
Oxycodone use is increasing
The use of oxycodone has been steadily increasing over the last three to four years (Figure 1).
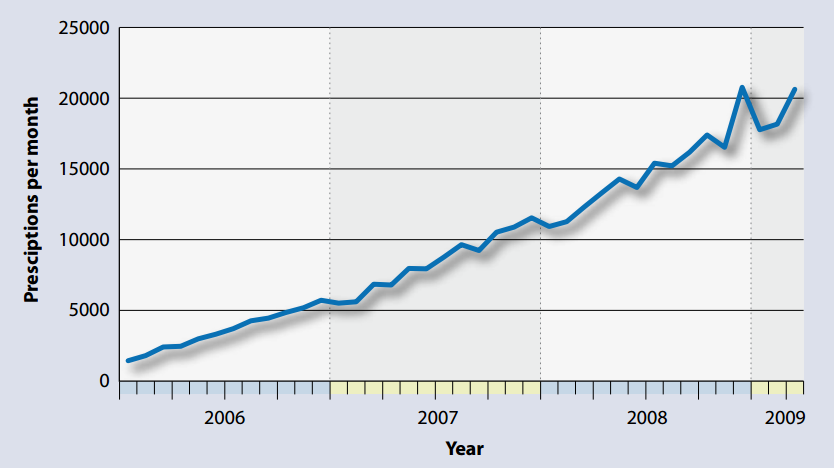
Figure 1: Number of oxycodone prescriptions in New Zealand 2006 to 2009 (Pharmaceutical
Warehouse data)
This trend is similar to patterns observed in other countries such as the UK and Australia and corresponds with a prominent
marketing campaign suggesting that oxycodone should be the preferred opioid analgesic for the treatment of moderate to
severe persistent pain. Oxycodone is more expensive than morphine, has a similar side effect profile and there is no clinical
evidence to support its use first-line.
Oxycodone is a strong opioid similar to morphine
Oxycodone is a semi-synthetic opioid with effects similar to morphine. It is an alternative to morphine for severe pain
at step three on the WHO analgesic ladder
 See BPJ 16, September 2008 “Pharmacological
management of chronic pain”.
See BPJ 16, September 2008 “Pharmacological
management of chronic pain”.
Clinical trials indicate that oxycodone is as effective as morphine at controlling cancer pain but with no significant
difference in overall tolerability. There is no evidence that oxycodone is superior to morphine for chronic, non-cancer
pain.
There is some evidence that there is individual variation in analgesic response and sensitivity to the adverse effects
of opioids.1 Oxycodone can be considered for the small number of patients who experience allergy or ongoing
neurotoxic adverse effects to morphine, such as hallucinations. Oxycodone may have a place in the management of complex
pain syndromes.
Oxycodone is not a substitute for codeine at step two on the analgesic ladder
One possible reason for the increased prescribing of oxycodone is that it is being used in place of codeine. From its
name (oxycodone) it may be perceived as being similar to codeine (a weak opioid), but in fact oxycodone is a strong opioid
twice as potent as morphine and with similar adverse affects. If pain is not controlled adequately with a step two analgesic,
including codeine, progress to step three might be indicated with morphine as the first line choice.
Precautions
Potential drug interactions
Oxycodone shares the same drug interactions as the other opioid analgesics. However, in contrast to morphine, the enzymes
CYP2D6 and CYP3A4 are involved in the hepatic metabolism of oxycodone, and there is the potential for drugs which inhibit
these enzymes (e.g. fluoxetine, erythromycin) to increase oxycodone plasma concentrations. There appears to be little
evidence at present that these potential interactions are clinically significant, but as with any drug it is important
to check the interaction profile prior to prescribing.
Adverse effects
Oxycodone is not a safer alternative to other opioid analgesics and it has the same spectrum of adverse effects. Of
greatest concern is the potential for respiratory depression when used at too high a dose or when combined with other
CNS depressants such as benzodiazepines and alcohol. Combination with other opioids is potentially lethal and care should
be taken to avoid this, especially if people might have access to a supply of both, e.g. after switching from morphine.
Dosage adjustment still required in renal impairment
Oxycodone has a better profile in renal impairment as, unlike morphine, it does not have active metabolites that are
renally excreted. However, caution is still required as the half-life of oxycodone is increased with renal impairment
and dosage adjustment is required.
Promotion of oxycodone
Oxycodone preparations have been heavily marketed over the last few years but advertisements do not provide useful
information to guide its rational and appropriate use.
- The wording in the advertisements strongly suggests to the prescriber that oxycodone is generally superior to other
opioid analgesics, but does not provide any evidence to support this.
- All statements supporting the use of oxycodone could be equally applied to morphine.
- When the trials that are cited to support the efficacy of oxycodone are analysed, it is found that they do not describe
any benefits over other opioid analgesics. For example, the statement “proven efficacy in providing long acting
relief from moderate to severe persistent pain” is supported by three references:
- A comparison between modified release and normal release oxycodone for chronic back pain (equally effective)2
- A controlled trial of oxycodone versus placebo for osteoarthritis pain (oxycodone superior to placebo)3
- A comparison of controlled release morphine with controlled release oxycodone in cancer pain (equally effective)4
- None of these trials indicate that oxycodone is anything other than an effective second-line alternative to morphine.
Prescribing oxycodone
The pharmacodynamics of morphine and oxycodone are comparable and equivalent formulations can be used in similar ways.
Oxycodone is available as:
- OxyNorm = normal release liquid (5 mg/5 mL) and capsules (5 mg/10 mg/20 mg)
- OxyContin = modified release tablets (5 mg/10 mg/20 mg/40 mg/80 mg). The modified release tablets have a biphasic
release profile, which gives an onset of analgesia within an hour of dosing and a duration of action of 12 hours
- Parenteral formulation (OxyNorm 10 mg/mL) for subcutaneous or intravenous injection or infusion. This is 1.5 to 2
times more potent than oral oxycodone.
Changing from oral morphine
Oral oxycodone is approximately twice as potent as oral morphine. To convert from oral morphine to oxycodone it is necessary
to halve the dose e.g. 10 mg morphine is equivalent to 5 mg of oxycodone. It is important to note that conversion rates
are an approximate guide and patient response should be carefully monitored.
Starting oxycodone in an opioid naïve patient
It is uncommon to commence oxycodone in an “opioid naïve” patient. One reason to consider this would be
in a “morphine phobic” patient who can be convinced to trial an opioid with a different name.
If opioid treatment with oxycodone is commenced, one possible approach is:
- Start with normal release formulation to find the daily dose required. As the half-life of the OxyNorm is slightly
longer than the half-life of morphine it may be dosed at six hourly rather than four hourly intervals.
Example of a prescription for an “opioid naïve” patient with normal renal function:
Oxynorm 2.5 mg to 5 mg four to six hourly
Plus
A = antiemetic e.g. haloperidol 1.5 mg nocte or as required for first five to seven
days
B = breakthrough Calculate the “as required” dose (prn) at 1/6th to 1/10th
of the 24 hour regular prescription. For example if taking Oxynorm 2.5 mg four hourly (= 24 hour dose of 15 mg), prescribe
prn dose of 2.5 mg (maximum one extra dose, two hourly)
C = constipation Start regular laxatives e.g. Laxsol one to two tablets twice daily
and alter the dose depending on effect
-
Titrate up. If requiring extra doses, or in pain, increase the regular and the prn dose by 30–50% by going from 2.5
mg to 5 mg, to 7.5 mg, to 10 mg, to 15 mg, to 20 mg, to 30 mg, to 40 mg etc.
- Once the dose is stable, and the pain controlled, add up all the doses required in a 24 hour period (regular and prn)
and divide by two to find the modified release twice daily dose.
For example Oxynorm 10 mg, six doses each day, which adds up to 60 mg, is equivalent to Oxycontin 30 mg twice daily.
Reassess:
A – Any nausea should be settled by the first week.
B – Always prescribe equivalent breakthrough e.g. if taking Oxycontin 30 mg twice daily, prescribe
Oxynorm 5 mg to 10 mg prn (maximum one extra dose, two hourly)
C – Remember to reassess bowel function and readjust laxative dose as appropriate
An alternative approach is to start with a low dose of modified release oxycodone (Oxycontin). This may be particularly
appropriate for benign chronic pain where the goal is not complete pain control but improvement in function. For example,
start with 5 mg every 12 hours. The dose can then be titrated to effect as necessary.
Key resource:
Christchurch Hospital Palliative Care Service. Palliative Care Guidelines. Canterbury District Health Board, 2009. Available
from:
www.cdhb.govt.nz/ (Accessed
September 2009).
References
- Cherny N, Ripamonto C, Pereira J, et al. Strategies to manage the adverse effects of oral morphine: an evidence based
report. J Clin Oncol 2001;19(9):2542-54.
- Hale M, Fleischmann R, Salzman R, et al. Efficacy and safety of controlled-release versus immediate-release oxycodone:
randomized, double-blind evaluation in patients with chronic back pain. Clin J Pain 1999;15(3):179-83.
- Roth S, Fleischmann R, Burch F, et al. Around-the-clock, controlled-release oxycodone therapy for osteoarthritis-related
pain. Arch Intern Med 2000;160(6):853-60.
- Mucci-LoRusso P, Berman B, Silberstein P, et al. Controlled release oxycodone compared with controlled release morphine
in the treatment of cancer pain: a randomized, double blind, parallel group study. Eur J Pain 1998;2(3):239-49.