Background
PCOS is characterised by a varied and often complex array of metabolic and endocrine abnormalities. The syndrome was
originally described by Stein and Leventhal in 1935, as a triad consisting of amennorhoea, hirsutism and obesity, in women
who had multiple cysts on their ovaries.1 Over the last decade or so, the understanding of this syndrome has
changed and the emphasis is often on the long-term consequences that may occur.
Prevalence of PCOS
PCOS is the most common endocrine disorder among young women. Accurate prevalence figures are hard to find because of
the lack of consensus that has existed regarding diagnosis,2 however it is reported to affect between 5-10%
of women of reproductive age.2, 3 New Zealand studies report a similar proportion of women with PCOS although
the ultrasound finding of polycystic ovaries is considerably more common, being found in 21% of randomly selected New
Zealand women.4 PCOS is also often undiagnosed.5
Cause of PCOS not fully understood
The pathogenesis of PCOS is not fully understood.6 There is some evidence of a polygenic component.7,
8 Insulin resistance is an important element in the development of PCOS but there are complex interactions involving
many systems (Figure 1).
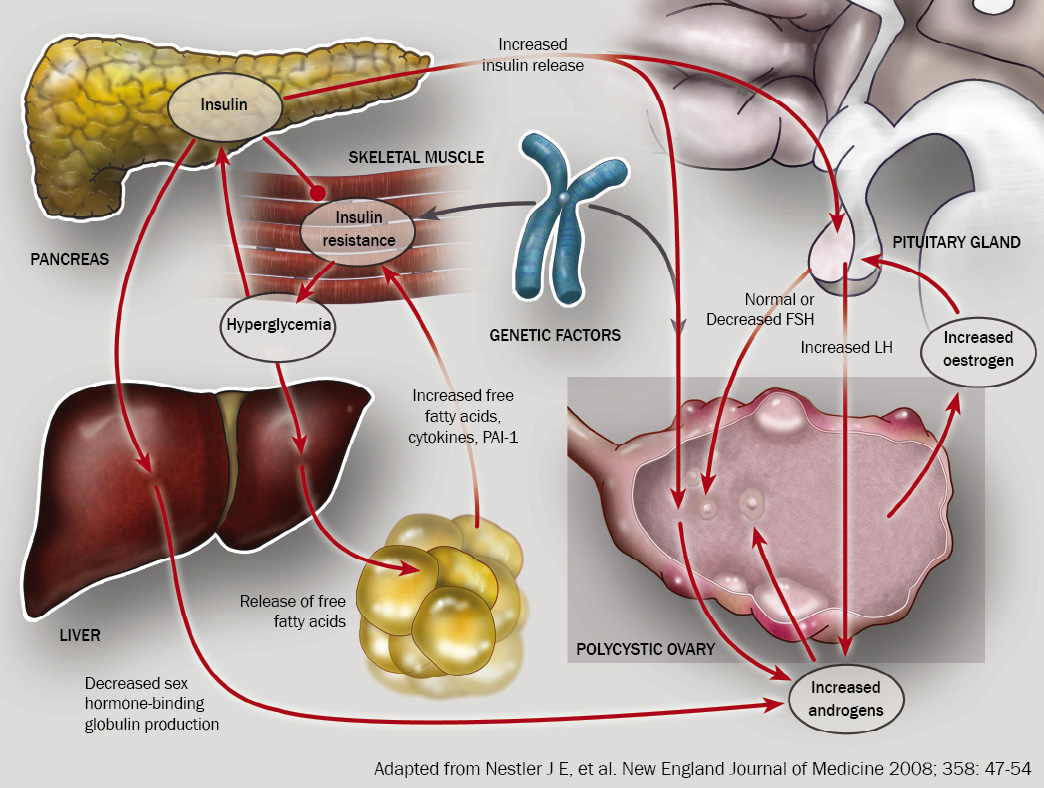 |
Figure 1: Pathophysiological characteristics of PCOS. This figure illustrates the complex interactions underlying
the pathophysiology of PCOS. Insulin resistance and the resulting hyperinsulinemia are responsible for the majority
of the changes found in PCOS. |
Long term health risks in PCOS
It is generally accepted that women with PCOS are at increased risk of:
- Impaired glucose tolerance,
- metabolic syndrome, gestational diabetes and type 2 diabetes9
- Hypertension, dyslipidaemia and cardiovascular disease10
- Fertility problems
- Endometrial hyperplasia and therefore endometrial cancer11, 12
Recent studies have shown an increased risk of obstructive sleep apnoea, irrespective of BMI.12 An association
between PCOS and breast and ovarian cancers has also been suggested but the evidence is conflicting.11, 13
Studies have identified that insulin resistance appears to be responsible for many of these long term health consequences.8,
14 Obesity contributes to the risks, but not all women with PCOS are obese. Hyperinsulinaemia and other metabolic
changes are present even in lean women with PCOS.11
There are, however, other factors often present in women with PCOS that may also contribute to these health risks. For
example, unopposed oestrogens are a risk factor for endometrial hyperplasia and carcinoma. In addition, both diabetes
and obesity have been linked to an increased risk of endometrial carcinoma.
| Box 1: Rotterdam Consensus on Diagnostic Criteria for PCOS9 |
Two out of three of the following:
| 1. |
Oligo- or anovulation |
| 2. |
Clinical and/or biochemical signs of hyperandrogenism* |
| 3. |
Polycystic ovaries** |
and exclusion of other etiologies (congenital adrenal hyperplasia, androgen-secreting tumours, Cushing�s
syndrome)
| * |
Hirsutism, acne, male pattern baldness, elevated total or free testosterone |
| ** |
On ultrasound, ?12 follicles of 2-9mm diameter and/or increased ovarian volume (>10mL) |
|
A woman with PCOS therefore may have many factors that could increase her long term health
risks and it has been difficult so far to determine the exact roles of each factor.13
Diagnosing PCOS
Diagnostic criteria have been developed for PCOS
PCOS is a syndrome, so there is no single diagnostic test. Diagnostic criteria have been developed (Box 1) and
widely adopted internationally. However, diagnosis can be difficult due to the variation in presenting symptoms and because
symptoms differ with age at presentation and change over time.
Presenting features of PCOS
Although presenting features (Box 2), age of presentation and severity of PCOS vary, a common presentation may be of
a woman with a history of gradually worsening hirsutism and irregular periods, which goes back for some years. For many
women however, failure to conceive may be the initial reason for presentation.
| Box 2: Presenting features of PCOS5, 8 , 13, 14 |
Presenting features (% affected)
| • |
Hyperandrogenism (hirsutism 70%, acne 30%, alopecia 10%, but not virilisation*) |
| • |
Menstrual disturbance 60-70% |
| • |
Infertility 70% |
| • |
Obesity, particularly truncal 35-50% |
| • |
Polycystic ovaries visible on ultrasound in asymptomatic woman 22-33% |
| • |
Acanthosis nigricans 1-3% ** |
| * |
Rapid development of virilisation signals a need for investigation to rule out the presence of an androgen secreting
tumour.8, 15 |
| ** |
A brown-discoloured �velvety� texture to the skin typically in the region of the axillae and the back of the neck,
often considered to be the cutaneous manifestation of insulin resistance (or hyperinsulinaemia). |
|
A full history is needed
It is important when taking the history to include questions about:
- Reproductive health (menarche, past and present cycle, oligo-/amenorrhoea, menorrhagia, miscarriage, infertility)
- Presence of androgenic symptoms (acne, hirsutism, alopecia of the scalp)
- Lifestyle factors (changes in body weight, eating and exercise habits, alcohol and smoking history)
- Family history of PCOS, diabetes, obesity, hirsutism and premature male baldness.13
Examination includes general as well as reproductive features
Examination of a woman with suspected PCOS should include an assessment of:
- Weight (both BMI and hip/waist ratio)
- Acne and hirsutism
- Blood pressure
Additional examination depending on the presenting features may include breast, abdominal and pelvic exam. The presence
of abdominal striae could indicate weight change or Cushing�s syndrome. A bimanual examination may identify ovarian enlargement,
although clinical pelvic examination is a poor predictor of polycystic ovaries, especially if the BMI is high. The presence
of features of virilisation (which may include frontal balding, deepening of the voice, broadening of the shoulders, breast
atrophy, clitoromegaly and loss of vaginal rugae) may raise concerns about other serious conditions.13
Investigation of PCOS
A clinical or biochemical finding of increased androgen levels along with either menstrual abnormalities or polycystic
ovaries on ultrasound will satisfy the current diagnostic criteria (Box 1). The initial tests recommended
for diagnosis are outlined in Table 1. While not essential for diagnosis, some clinicians still suggest testing LH/FSH.
Once the diagnosis is established, fasting glucose and lipids are recommended. Other tests may be required depending on
clinical suspicion, to exclude other conditions (Table 2).
Table 1: Recommended investigations for aiding diagnosis of PCOS
| Investigation |
Expected finding in PCOS |
Comment |
| Recommended investigations for diagnosis |
| Exclude pregnancy |
|
Most common cause of amenorrhoea.13, 14 |
| Pelvic ultrasound |
Polycystic ovaries |
Important as part of the diagnostic criteria but not a “must
do” if diagnosis is made on clinical and biochemical grounds. |
| Free testosterone |
Usually increased |
More sensitive for identifying physiologically active androgens. This
is calculated from total testosterone and SHBG. Very high levels of total testosterone require further investigation
to rule out other causes such as late-onset congenital adrenal hyperplasia, Cushing�s syndrome, adrenal or ovarian
tumour. SHBG levels are decreased in PCOS. |
| Recommended investigations after diagnosis |
| Glucose |
|
To check for glucose intolerance or diabetes. If fasting level > 5.5
mmol/L or random > 7.7 mmol/L then a glucose tolerance test is recommended |
Lipids
|
Usually high triglycerides, lower HDL and mildly elevated LDL.11 |
A fasting level may be useful in establishing cardiovascular risk. |
| Other tests to consider |
LH/FSH
|
LH will often be increased,
FSH usually normal,
giving an increased ratio |
While not essential for diagnosis, some clinicians remain convinced
of the value of LH testing in predicting future complications of PCOS. |
Table 2: Tests to exclude other conditions (depending on clinical suspicion)
| Test |
Reason |
Clinical signs |
| Prolactin |
Very high levels may suggest a pituitary cause or medication use (especially antipsychotic
medication) |
Galactorrhoea
Irregular or absent periods |
| TSH |
To exclude thyroid abnormalities as a cause of menstrual irregularity14 |
Menstrual changes associated with other thyroid symptoms (either hypo or hyper) |
| Oestradiol + FSH |
To help exclude premature ovarian failure (low oestradiol, very high FSH)7 |
Menopausal symptoms and signs in women less than 40 years |
| 17-OH progesterone |
To help exclude late-onset or non-classic congenital adrenal hyperplasia (very rare) |
Difficult to distinguish clinically from PCOS. However there may be a family history
of CAH, less menstrual disruption or history of early growth of pubic hair |
| DHEAS |
A marker for adrenal androgen production, very high levels may be associated with an
adrenocortical tumour15 |
Rapid onset of virilising features |
| Androstenedione |
A marker for ovarian androgen production, very high levels may be associated with an
ovarian androgen secreting tumour15 |
Rapid onset of virilising features |
| 24hr urine cortisol |
Increased in Cushing�s syndrome |
Typical Cushingoid features e.g. central obesity, moon face, thinning of skin, striae,
excessive sweating |
Treatment and management of PCOS
| Changes in serum endocrinology in PCOS8 |
There are multiple biochemical changes in women with PCOS. The key feature is the
increased level of serum androgens which are responsible for most of the common presenting features:
- Increased androgens (testosterone, androstenedione and dehydroepiandrosterone sulphate (DHEAS))
- Increased luteinising hormone (LH)
- Decreased sex hormone binding globulin (SHBG)
- Increased prolactin
- Increased oestradiol
- Increased insulin
|
Lifestyle modification to reduce weight is the most effective first line treatment in PCOS.14,
16, 17 Even a modest weight loss of 5% will reduce central obesity and insulin resistance and improve endocrinological
abnormalities and menstrual irregularity (including increasing the rate of ovulation).17 Ultimately, women
who succeed in losing weight are more likely to achieve and have a healthier pregnancy and reduce their risk of gestational
diabetes. Longer term benefits of weight loss result from the reduction in insulin resistance. Note that weight loss
is not necessary if BMI is within normal range.
Treatment may be required for acne and hirsutism, which are often the major reasons for women to present.
Treatment options may include anti-androgens, topical agents (particularly for acne) and local hirsutism treatments (including
electrolysis and laser therapy). The combination of acne, hirsutism and obesity is likely to lower self esteem in women
with PCOS. Psychological support may be required and this may also help women achieve the recommended beneficial lifestyle
changes.
| Do not test insulin |
| Fasting serum insulin is a poor measure of insulin resistance.18 Although
used widely in large population-based epidemiological studies, it is not recommended for use in a general practice setting.
It is more useful to identify the risk factors that are associated with insulin resistance (and often therefore identify
metabolic syndrome and PCOS). These risk factors include raised fasting glucose and lipid levels, high blood pressure
and central obesity. |
First line anti-androgenic therapy is often in the form of a combined oral contraceptive pill containing
cyproterone acetate, and/or the diuretic spironolactone (usually 100�200 mg/day), which has an anti-androgenic effect.
As a second line treatment, higher dose regimens of cyproterone acetate or spironolactone may be combined with oral contraceptive
pill use.
Regulation of the menstrual cycle may be achieved with weight loss, a combined oral contraceptive or
progesterone therapy (if COC not tolerated). Most clinicians would currently recommend the use of these hormonal treatments
to protect the endometrium from unopposed oestrogen stimulation in women who have chronic anovulation.11, 14
Metformin, which is an insulin sensitising agent, has been advocated as a treatment for PCOS. Theoretically
it should decrease insulin levels and therefore reduce androgen production, and help restore the endocrinological abnormalities
of PCOS. It has been suggested that it may aid weight loss, but there is currently no evidence to support this.14
There is ongoing debate regarding the appropriateness of metformin as first choice treatment in women with PCOS who
are having fertility problems. It appears that in many studies metformin results in no improvement in live birth rates
compared to clomiphene citrate.18, 20 A multi-centre New Zealand randomised trial PCOSMIC (PCOS Metformin for
Infertility with Clomiphene) will help to define the place of metformin in ovulation induction and is expected to be completed
in 2008.21
| Ethnicity and PCOS |
| Limited data exist on prevalence between different ethnic groups in New Zealand.
A cross-sectional study of women presenting to the gynae-endocrine clinic at National Women�s Hospital who were diagnosed
with PCOS showed rates for European, Māori and Pacific Island women in proportion to the general population.
Although numbers were small, Indian women appeared to be over-represented and Chinese women under-represented. What
may be more important though is that Māori and Pacific Island women with PCOS were more likely to be obese and
had significantly more adverse metabolic features, higher levels of androgens, triglycerides, LDL cholesterol, fasting
insulin, systolic and diastolic blood pressure, and lower HDL.19 |
If infertility is the main presenting problem, specialist referral is recommended. Clomiphene citrate
is considered first line treatment.14, 22 To avoid the risk of over-response leading to multiple pregnancy,
clomiphene citrate treatment is carefully monitored through a fertility clinic (with late follicular serum oestradiol
levels and ultrasound scanning when appropriate). Weight reduction, if appropriate, remains central to the success of
any treatment.
Ongoing preventive screening of cardiovascular and endometrial disease risk factors is important when
managing women with PCOS. There are no consensus guidelines in widespread use. A sensible approach would be to check BMI
and blood pressure annually, along with fasting lipids and a glucose tolerance test every three to five years in patients
with low cardiovascular risk, or every one to three years where other risk factors, such as obesity, are present.
These consultations give women with PCOS the opportunity to review lifestyle factors to optimise their long term health.
For women with anovulation who elect not to use endometrial protection, regular screening by transvaginal ultrasound and/or
endometrial biopsy every one to two years is advisable.