This report presents how testing for diabetes has changed since the recommendations were published. Patients with diagnosed diabetes were excluded from the analysis using a virtual diabetes registry algorithm that included diabetes medicines, laboratory tests and hospital discharges for diabetes-related diagnosis codes. This algorithm identified 186,560 registered patients with diabetes (4.4% of all registered patients). The data in this report was sourced from the Ministry of Health National Collections.
In this report 
View / Download pdf version of this report
In September 2011, the New Zealand Society for the Study of Diabetes (NZSSD) issued a position statement recommending
the use of glycated haemoglobin (HbA1c) for the diagnosis of type 2 diabetes. 1
- HbA1c is now the recommended diagnostic test for type 2 diabetes
- Fasting plasma glucose remains a useful test in patients where HbA1c cannot be used
This report will discuss how testing patterns have changed since the recommendations were released, including data for
all registered New Zealand patients, and feedback on the testing received by your practice's registered patient population.
Which test should I use to diagnose type 2 diabetes?
In September 2011 HbA1c replaced fasting plasma glucose as the recommended test for diagnosis and testing for type 2
diabetes in most patients. This report presents how testing for diabetes has changed since the recommendations were published.
Patients with diagnosed diabetes were excluded from the analysis using a virtual diabetes registry algorithm that included
diabetes medicines, laboratory tests and hospital discharges for diabetes-related diagnosis codes. This algorithm identified
186,560 registered patients with diabetes (4.4% of all registered patients). The data in this report was sourced from
the Ministry of Health National Collections.
| When to use HbA1c |
When to use fasting plasma glucose |
HbA1c should be the test of choice for testing for and diagnosis of type 2 diabetes in most people.
HbA1c has a number of advantages including:
- There is no need for fasting and there are simpler sampling and analysis requirements
- It is less affected by day to day variation in glycaemia, and there is less biological variation than with fasting
glucose
- It measures chronic glycaemic exposure rather than an acute value
|
Fasting plasma glucose remains a valid test for diagnosis of type 2 diabetes in situations where HbA1c cannot
be used, for example, in patients with specific conditions which may lead to an inaccurate HbA1c level.
In women who are pregnant, the recommendation is to continue to use oral glucose tolerance testing (75 g) for the
diagnosis of gestational diabetes in women with an abnormal polycose screen (50 g). |
Has testing activity changed in line with the new recommendations?
National
Data: Pattern of tests requested per month for patients without diagnosed diabetes » 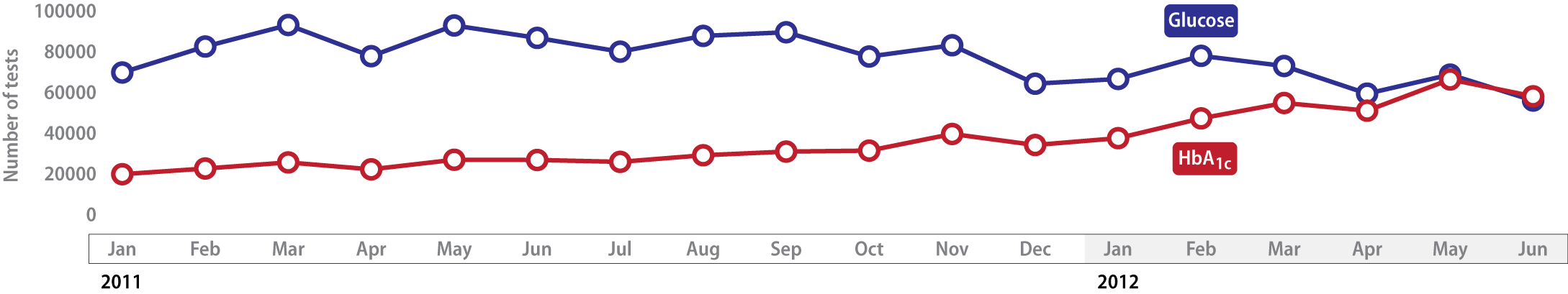
In the nine months following the change in recommendations, HbA1c testing for people without diagnosed diabetes increased
by 98% compared with the nine months before, while glucose testing decreased by only 16%. Some practitioners may be requesting
both tests, which is unnecessary in most cases. We would expect glucose testing in patients without diagnosed diabetes
to continue to decline until there are many more HbA1c tests than glucose tests.
In the section below the testing for diabetes before and after recommendations changed is presented as a rate per 1000
patients, this will allow you to compare the rate of testing in your registered patients with the national rate.
Click
to enlarge image chart »
- New Zealand Society for the Study of Diabetes. NZSSD position statement on the diagnosis of, and screening for type 2 diabetes. 2011. Available from:
www.nzssd.org.nz (Accessed October, 2012).
- National data excludes tests for MidCentral, Nelson Marlborough, South Canterbury, Tairawhiti,
Wairarapa and Whanganui DHB areas (representing 11.5% of all NZ patients) due to incomplete recording
of HbA1c tests after the change to reporting results as mmol/L instead of % which coincided
with the release of these guidelines. Where your practice data may be affected, this is indicated on the report.