View / Download
pdf version of this correspondence
A weed
from far away
Dear bpac,
Thank you for your article on Māori Health. It surprises me that it did not mention cannabis which in my opinion
is one of the major factors underlying adverse physical and mental health of people who use it, and it reduces the
individuals motivation to strive for good health.
I did your recommended search of my high risk Māori males >35, which was a very valuable and instructive exercise.
Analysis of their notes revealed that most of them either smoke or have been smokers, and at least 33% of them smoke
cannabis. Cannabis tends to induce a mental state of apathy, and it is not surprising that cannabis smokers are not
highly motivated to improve their health while continuing to smoke it.
Why did your article not say anything about this extremely important adverse influence on Māori health?
We will never get to the root of ill health if we chose to ignore it.
He Taru Tawhiti - "a weed from far away"
GP, South Island |
We agree that cannabis use is an important health issue for all communities and we will consider addressing this in
a future edition of BPJ.
BPJ 13
Māori Health Edition - Immunisation
Dear bpac,
As a Pakeha having worked as a Tamariki Ora nurse at Whaiora Whanui Māori health provider in Masterton and Arai
Te Uru Whare Hauora in Dunedin I am heartened to see Best Practice magazine acknowledge the diverse reality of Māori
people.
I read with interest the item on bronchiectasis as I had not connected that disease with immunisation before. I noticed
the statistics for Māori two year olds being fully immunised had increased significantly since the referenced
figure of 42%. As of 24th April 2008, according to the National Immunisation Register, the national immunisation coverage
rate for Māori at age two years is 68%, with a variance across the 21 DHBs between 56% and 90%. All 21 DHBs now
have childhood immunisation outreach services and general practices are working hard to reduce disparity, going the
extra mile to recall or refer those children who might otherwise miss out.
Our message in general practice is to encourage access to timely immunisation to avoid breakthrough disease, in particular
from Hib, Pertussis and Strep. pneumoniae. On Page 26 of
BPJ
13 I read the sentence ; 'Rheumatic fever is unlikely to be seen in children under three years because their immune
systems are not fully developed'. I would prefer to see the terms 'inexperienced' or 'naive' used in this context to
avoid perpetuating the misperception that we should wait until children are older before immunising. Trained vaccinators
are aware that the infant immune system is fully developed at birth and that the vaccines on the immunisation schedule
in New Zealand make a miniscule contribution to the massive antigenic challenges in the natural environment. All of
our communication ought to convey this message on the importance of timeliness as the alternative delaying immunisation
leaves us open to criticism in the event of an unprotected child becoming ill from a vaccine preventable disease.
Kind regards
Barbara Warren, Coastal Otago Immunisation Coordinator |
Amiodarone
monitoring
Dear bpac
In BPJ 2, December 2006, you ran an article on amiodarone
monitoring requirements. The article suggested a range of tests that are not practical in a GP practice, for patients
or GPs to manage. The article finishes with an editors note to revisit the requirements in the next issue - I cannot
find any follow up.
GP, Waikato |
We asked Cardiologist, Dr Stewart Mann to comment on the issue of monitoring patients on amiodarone.
He says:
As a recent paper1 suggested, many monitoring recommendations are not evidence based and some recommendations
are over the top pragmatically with no evidence of more rigorous monitoring leading to fewer important side effects. For
example, the usefulness of any routine monitoring of pulmonary function tests or chest x-ray to pick up early fibrosis
is not at all established. Some clinicians suggest more intensive monitoring may be justified with higher amiodarone maintenance
doses (400mg/day +).
Reasonably pragmatic guidelines produced in 20002 suggest a number of baseline tests and follow-up clinical assessment
(history and examination) every three months in the first year (Table 1).
It is likely that a cardiologist would follow a patient newly prescribed amiodarone three-monthly for the first year
and be responsible for the recommended tests over that period. GPs may take responsibility thereafter if no cardiac follow-up
is otherwise required.
Table 1: Recommended amiodarone monitoring
| |
Baseline |
Follow up |
| 6 monthly |
Annually |
| Electrocardiogram (ECG) |
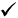 |
|
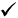 |
| Chest x-ray (CXR) |
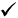 |
|
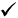 |
| Thyroid function tests (TFTs) |
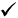 |
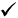 |
|
| Liver function tests (LFTs) |
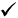 |
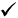 |
|
| Pulmonary function tests (PFTs) |
Only if any symptoms of respiratory deficiency |
Only for those with suspicious symptoms |
| Eye examination |
Only if visual impairment |
Slit lamp assessment suggested for those with suspicious symptoms |
Reference:
- Stelfox H, Ahmed S, Fiskio J, Bates D. Monitoring amidodarones toxicities: recommendations evidence and clinical practice.
Clin Pharmacol Therap 2004:75(1):110-22.
- Goldschlager N, Epstein AE, Naccarelli G, et al. Practical guidelines for clinicians who treat patients with amiodarone.
Arch Intern Med; 160(12): 1741-8.
The use
of SSRI's for treating depression: An Open letter
In February 2008, researchers at the University of Hull, UK, published an article which concluded that "there seems
little evidence to support the prescription of antidepressant medication to any but the most severely depressed patients".
[Kirsch I, Deacon BJ, Huedo-Medina TB et al. Initial severity and antidepressant benefits: a meta-analysis
of data submitted to the Food and Drug Administration. PLoS Med 2008;5(2):e45].
The following day, New Zealand newspapers ran the story; "Anti-depression drugs don't work".
In March 2008, the BMJ published an article by Turner and Rosenthal [BMJ 2008; 336-:516-7]. Turner and Rosenthal
respond to the conclusions of Kirsch et al and point out that the efficacy of antidepressants is not an absolute measure
but depends on how clinical significance is defined.
In an earlier article (January 2008), Turner and Rosenthal assessed FDA data on 12 antidepressant drugs, and derived
an overall effect size of 0.31 (on a scale from 0-1). Kirsch et al used the same FDA data to examine four of these
12 drugs and calculated an overall effect size of 0.32. Despite the apparent agreement, interpretation of these results,
by Kirsch et al on the one hand and by Turner and Rosenthal on the other, has been quite different. In contrast to
the conclusion of Kirsch et al that antidepressants are ineffective, Turner and Rosenthal concluded that each drug
is superior to placebo.
Turner and Rosenthal explain their different interpretation in relation to Kirsch's use of the criteria for clinical
significance recommended by NICE. Clinical significance is important because drug trials can show benefit of drug over
placebo that is statistically, but not clinically, significant. Tests of statistical significance (e.g. p values) tell
whether the true effect size is zero or not (i.e. they use the null or no effect or no difference hypothesis) but say
nothing about the size of the effect. Effect size is required to gauge clinical significance. By convention, values
of 0.2, 0.5 and 0.8 represent small, medium and large effects respectively - the values chosen are relative (to each
other) and they are arbitrary - i.e. they are no more reliable than intuition. Using this convention, NICE chose 0.5
as the cut-off below which they deem drug benefit to be not clinically significant. But this decision transforms a
continuous measure of effect into an all or none (yes/no) measure and Kirsch's (fallacial) conclusion that the overall
statistical effect size of 0.32 equates with NO benefit from antidepressant drugs.
Actually, 0.3 is a positive effect - not a full effect, but a significant effect between small and medium. Given
this clinical interpretation of effect size, Turner and Rosenthal advise a circumspect but not dismissive approach
to use of antidepressants - patient response is not all or none; partial response should be expected.
In the light of this appraisal of the original report by Kirsch et al, we suggest that GPs should take the decision
to prescribe or not prescribe SSRI treatment, for patients with mild to moderate depressive illness, on a case-by-case
basis.
Dr Peter Cardon, Chair of the South Link Health Clinical Advisory Committee |
Gastroprotection
for people taking NSAIDs?
Dear bpac,
For patients who need long term NSAIDs (and we try to keep this to a minimum!) do you still recommend
omeprazole 20mg/d (or equivalent PPI). Would ranitidine 300mg/d be an alternative?
Also do you recommend checking for H. Pylori (by stool) first before starting long term NSAIDs?
Is there any good evidence for the above or is it just expert opinion?
Thank You,
GP Peer Review Group, Nelson |
NSAIDs can cause several problems in the gut such as bleeding, perforation, ulceration and dyspepsia.
The first thing to consider is minimising risk for all people about to start long-term NSAIDs.1 For example,
factors to consider are:
- Is there a safer alternative (regular paracetamol)?
- Can you choose an NSAID with a lower relative risk of GI toxicity, e.g. ibuprofen?
- Is the lowest effective dose being used?
- Has the person been informed of potential adverse effects and what to do should they occur?
- How long does the treatment need to continue? Regular review of risk status is needed.
- Is the person on aspirin? If so:
- Has the additive effect of aspirin and any other NSAID been considered?
- Is the person on a COX-2 selective drug? COX-2 drugs lose their selectivity in the presence of aspirin.
| The following characteristics predict the risk of adverse upper GI events in those taking NSAIDs:
Use of NSAID (includes aspirin, and COX-2 plus aspirin) plus any of the following:
- History of peptic ulcer
- History of upper GI bleeding
- Concomitant disease, especially coronary heart disease
- Increased frailty such as substantial arthritis-related disorder
- Previous NSAID gastropathy
- Concomitant use of corticosteroids, anticoagulants or bisphosphonates
- High dose of NSAID (includes NSAID + aspirin)
- H. pylori infection
|
Co-prescription of agents that protect the gastrointestinal mucosa is recommended for those aged 65 years or over with
one additional risk factor and for those aged less than 65 years with two or more risk factors (see box).1
Proton pump inhibitors (omeprazole 20mg/day or equivalent), or misoprostol (≥ 600mg/day if tolerated) may be considered
for protection against NSAID-associated gastric or duodenal ulcers. Adverse effects such as colic and diarrhoea may limit
the use of misoprostol.
H2-receptor antagonists may not be adequate for NSAID gastro-protection against the more severe complications but they
do improve dyspepsia related to NSAID use.
Consider cardiovascular risk also
It is important to consider an individual's cardiovascular as well as gastrointestinal risk, before initiating
any NSAID (especially a COX-2), and then to consider the questions of - with or without a gastroprotective agent, and
with or without concomitant aspirin? See Table 1.
Table 1. Prescribing NSAIDs for people with cardiovascular or gastrointestinal risks
| |
No or low NSAID GI risk |
NSAID GI risk |
| No CV risk (without aspirin) |
Non-selective NSAID |
COX-2 or non-selective NSAID + PPI COX-2 + PPI for prior GI bleeding |
CV risk
(with aspirin) |
Naproxen* + PPI if GI risk of aspirin/NSAID combination warrants gastroprotection |
PPI irrespective of NSAID. Naproxen if CV > GI risk COX-2 +PPI for prior GI bleeding |
* Naproxen is not associated with an excess risk of vascular events as are some other traditional NSAIDs
NSAID use and H. pylori infection are independently associated with an increased risk of gastrointestinal bleeding and
ulceration. Treat known H. pylori infection in people about to start long-term NSAID therapy.
In people who are already taking an NSAID, eradication of H. pylori is unlikely to reduce the risk of NSAID-induced
bleeding or ulceration.
In New Zealand H. pylori prevalence is generally low so routinely testing for it pre-NSAID is probably not
worth it, particularly in those who are not at high GI risk.
However patients about to start long term NSAID therapy who have a history of GI complications may benefit from testing
and eradication of H. pylori before initiation of treatment to prevent GI complications.2, 3
Thank you to Dr John Wyeth, Gastroenterologist and Clinical Leader, Capital and Coast DHB, for
his contribution to this answer.
 For more information on the risk of GI damage in NSAID use, see "Non-steroidal anti-inflammatory drugs (NSAIDs): Making safer treatment choices", BPJ 55 (October, 2013).
For more information on the risk of GI damage in NSAID use, see "Non-steroidal anti-inflammatory drugs (NSAIDs): Making safer treatment choices", BPJ 55 (October, 2013).
References:
- New Zealand Guidelines Group. Management of dyspepsia and heartburn, 2004. Available from:
http://www.nzgg.org.nz/ (Accessed
May 2008)
- British National Formulary (BNF). BMJ Publishing Group and Royal Pharmaceutical Society of Great Britain. March 2008.
- Ables AZ, Simon I, Melton ER. Update on Helicobacter pylori treatment. Am Fam Physician 2007; 75(3): 351-358.

Send your letters to...
Correspondence, PO Box 6032, Dunedin, 9059
or email editor@bpac.org.nz