In this article 
View / Download
pdf version of this article
Key points
- Sexually transmitted infections including Chlamydia and Gonococcal infections are increasing in New Zealand with
young people being most at risk
- Chlamydia testing should be considered in all sexually active patients under 25 years of age when they access healthcare
- Self taken vaginal swabs for women or first void urines for men can be useful for Chlamydia testing
|
This is the second of a series of articles focusing on sexual health. In the recent best practice journal, we examined
the prevalence of STIs in New Zealand.
This article provides an overview of some infections affecting the genital area, including some non-sexually transmitted
infections. All testing is guided by sexual history and genital examination.
The next best practice journal will finish this series, with articles about the treatment of STIs, and talking about
safer sex to patients.
Chlamydia trachomatis - still most common

Background: Chlamydia infection is the most frequently diagnosed sexually transmitted infection (STI)
in New Zealand and is most common in sexually active adolescents and young adults. The 2007 annual ESR STI surveillance1 reported
that the number of Chlamydia cases diagnosed had increased by 19.4% in the previous four years.
Transmission: Chlamydia is transmitted through contact with genital secretions resulting in infection
of the endocervix, urethra, rectum and occasionally pharynx and eye.
The most accurate results will be obtained at least 1-2 weeks after potential exposure due to an incubation period of
approximately 7-21 days. Swabs taken too soon after exposure may give falsely negative results.
Vertical transmission from an infected mother may cause conjunctivitis/ophthalmia in the newborn, this tends to occur
within three weeks of delivery.
Symptoms: Chlamydia is asymptomatic in approximately 70% of people.
Symptomatic men may present with urethral discharge (mucopurulent, milky or clear), urethral irritation, dysuria or
testicular pain/swelling. On examination there may be testicular tenderness.
Symptomatic women can present with vaginal discharge (mucopurulent, milky or clear), lower abdominal pain, dyspareunia,
dysuria or irregular PV bleeding. On examination, the cervix and lower abdomen may be tender.
Tip: Chlamydia testing should be considered in all patients found to have a sterile pyuria, as well
as women presenting with breakthrough bleeding while taking the combined oral contraceptive pill.
Testing: The Ministry of Health recommends that Chlamydia testing is considered in all sexually active
patients under 25 years of age,2 when they access healthcare whether or not they have symptoms.
Recommendations for Chlamydia Testing in New Zealand2
Recommendations for asymptomatic opportunistic testing
This is particularly important if the individual has not consistently used condoms.
Females
Testing should be offered to all sexually active females under 25 years of age if they have never been tested.
The offer of testing should be repeated annually to all sexually active females under 25 years of age if they have:
- had two or more partners in the last 12 months, or
- had a recent partner change.
Males
Consider testing in sexually active males if they are:
- aged under 25, and
- two or more sexual partners in the last year or a recent partner change, or
- co-infection with another STI
Recommendation for when Chlamydia testing is indicated

Testing should be routinely given to:
- those with symptoms suggestive of Chlamydia infection
- sexual partners of those with suspected or confirmed Chlamydia infection
- patients requesting a sexual health check
- patients with another STI
- pregnant women (test in first trimester and repeat in third trimester if there are ongoing risk factors)
- women undergoing a termination of pregnancy
- mothers of infants with chlamydial conjunctivitis or pneumonitis
- pre-menopausal women undergoing uterine instrumentation
- semen and egg donors
- men who have sex with men.
Over recent years more sensitive methods of Chlamydia testing have been developed. These methods include polymerase
chain reactions (PCR) which results in Nucleic acid amplications (NAA). This process is so sensitive that Chlamydia can
be detected using samples from anywhere in the vagina, leading to the advent of self-taken swabs being practical. First
void urine tests for men and self-taken vaginal swabs for women can be ideal for offering Chlamydia testing to asymptomatic
individuals as they are generally well accepted by patients.
First Void Urine (FVU) tests for men are generally as sensitive in detecting Chlamydia infection as
swabs and are generally better tolerated than traditional deep urethral swabs. This test is suitable for men with or without
symptoms but it is not recommended for first-line testing in women due to reduced sensitivity.3
In women the sensitivity of a self-taken lower vaginal PCR Chlamydia swab is comparable to a clinician
obtained endocervical swab4 and is generally preferred by patients.5,6 If the woman is undergoing
a speculum examination, a cervical swab is recommended.2
Taking a vaginal or endocervical swab for Chlamydia gives the best chance of detecting Chlamydia as the vagina is usually
the primary site of infection. Very occasionally it may be necessary to check for Chlamydia in the bladder as it can be
a cause of UTIs. If this is suspected and the vaginal swab comes back negative, a first void urine may be the most practical
thing to do, as this has better patient acceptance than urethral swabs.
Lymphogranuloma venerum
Another presentation of Chlamydia trachomatis is lymphogranuloma venerum (LGV) which can cause small
painless genital ulcers and subsequent painful chronic granulomatous inguinal lymphadenopathy.
It has historically been thought of as a “tropical disease” but in 2003 it was detected in Europe amongst
MSM (men who have sex with men)7 and by 2005 in Australia. The first confirmed case in New Zealand was in 2007.
Any patient with symptomatic anorectal disease, positive for Chlamydia and not responding to normal therapy, should
have LGV considered as a differential diagnosis.
LGV is diagnosed by rectal swab (for NAA technique) and by excluding other causes of enlarged lymph nodes or genital
ulcers.
| Instructions for collection of first void urine |
Instructions for self-taken lower vaginal PCR Chlamydia swab |
| A first void urine (FVU) is the first 25-30 ml of urine voided, preferably two hours since the man has
last urinated. If a man has recently voided urine, a deep urethral swab (5-6cm deep) may be performed instead, but most
men would probably choose to wait and collect the FVU after two hours have lapsed. |
The women should find a comfortable position, either sitting or standing and then insert the Chlamydia
swab at least 2-3 cm into the vagina (about half way along the swab). The swab should be turned around once within the
vagina and left for at least 10 seconds. Then the swab should be placed back into the tube. |
Gonorrhoea - Co-infection with Chlamydia is common

Background: In the last five years the number of Gonorrhoea cases in New Zealand has increased by over
50%.1 Co-infection with Chlamydia is common. Gonococcal infections rates are higher among young adults and
among men who have sex with men.
Transmission: Infection due to Neisseria gonorrhoeae is through contact with genital secretions and
like Chlamydia can result in infection of the endocervix, urethra, rectum and occasionally pharynx and eye.
The incubation period is approximately 2-5 days (range 2-10 days), so swabs taken too soon after exposure may be falsely
negative. Swabs will be more accurate if delayed until 5-7 days after potential exposure.
Vertical transmission from an infected mother may cause conjunctivitis/ophthalmia in up to 50 per cent of exposed neonates.
This tends to occur three to five days after exposure and is often more severe than other causes of ophthalmia neonatorum
(eg, chlamydia).
Symptoms: Infection can cause dysuria, vaginal discharge or lower abdominal pain in females and urethral
discharge (often mucopurulent) in men. It is asymptomatic in approximately 50% of females and 5% of males.1
Testing: In contrast to Chlamydia, swabs need to be taken from the endocervix in women, urethra in
men (1-2 cm deep), and rectum/pharynx if appropriate in either gender.
It is important that it is transported to the laboratory as soon as possible, as there is up to a 50% loss of viable
organism if there is over 24 hours before the sample gets to the laboratory.
Syphilis - on the increase

Background: Since 2002 there has been an increase in the number of people presenting to sexual health
clinics with syphilis after being relatively rare, in New Zealand, for decades previously.8 Rates have continued
to rise since.9 Although the overall number of cases of syphilis in New Zealand remains low compared to other
STIs, case numbers have more than doubled in the past five years.1
In New Zealand sexual health clinics the highest number of syphilis cases were found in MSM.9
Transmission: Treponema pallidum can enter the body by sexual contact through the skin lining the genital
tract, anus or mouth.
The incubation period is approximately three weeks.
The first stage is highly infectious and usually presents as a solitary painless ulcer (chancre) at
the site of infection which persists for between 10-100 days.
The second stage occurs if the primary stage is untreated and is also infectious. It begins approximately
2-8 weeks after the initial infection, when a rash may appear for 1-2 months, commonly on the hands/soles as well as the
rest of the body.
Third stage syphilis occurs in about one third of cases some months-years after the ulcer and rash
have disappeared, and can involve damage to the brain, heart or spinal cord.
Testing: At the early stages, exudate from the ulcer, transferred onto a glass slide, can be examined
by darkground microscopy in some laboratories.
Serological tests may include RPR, VDRL, and TPHA. Results may be negative for the first few weeks after the appearance
of the chancre although 90% seroconvert by six weeks after the appearance of the chancre and 100% by twelve weeks. Serology
can be repeated if initially non-reactive.
Note: In regions where non-venereal treponematosis (e.g yaws) are endemic such as in the Pacific (particularly
in Samoa), serological tests should be interpreted with care as these conditions can produce false positive tests.
Genital herpes - asymptomatic transmission is common

Background: Genital herpes infection is caused by Herpes Simplex Virus (HSV) types 1 or 2. Traditionally
HSV-1 was the usual cause of orolabial herpes (lips and mouth), while HSV-2 was the usual cause of genital herpes but
increasingly there is cross-over occurring. Periodic reactivation is a feature of this infection but HSV-2 recurs six
times more frequently than type 1.10
Transmission: HSV is spread by direct skin to skin contact. Viral shedding occurs most easily when
a sore is present but can occur even when a person has no symptoms. Most new infections are transmitted via asymptomatic
viral shedding.10
Symptoms: HSV genital infection may be asymptomatic or may cause painful genital lesions and sometimes
a discharge. Only 20% people with gential herpes has a typical syndrome, 20% are asymptomatic and the further 60% are
atypical symptomatic.
Testing: Swabs for viral typing, to differentiate between HSV-1 and HSV-2, should be obtained in all
patients with newly diagnosed genital herpes, as the information can influence prognosis and counseling.
Viral swabs are taken from the genital lesion (de-roof any vesicles if necessary) and broken off into viral transport
medium while awaiting transportation to the laboratory.
Type specific HSV serology is rarely clinically useful, although may sometimes be useful for people with a history of
genital symptoms but have never had a successful confirmatory test, or a pregnant woman presenting with her first clinical
episode of herpes while she is pregnant. It takes several weeks for an antibody response to occur following the primary
exposure to HSV.
Trichomoniasis

Up to 50% of men and women infected with Trichomonas vaginalis will be asymptomatic.
After exposure, symptoms take approximately four weeks to develop. In women this can include dysuria, vaginal irritation
and sometimes a yellow-green frothy vaginal discharge. Men may experience a thin milky discharge and dysuria. The normal
incubation period is 4-28 days. Testing is with a high vaginal or male urethral swab.
Bacterial vaginosis (BV) - also referred to as gardnerella - females only
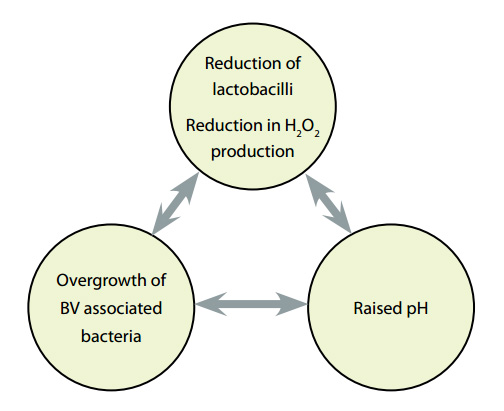
Background: The exact mechanism for the onset of BV remains a mystery. It is associated with a reduction
in lactobacilli and hydrogen peroxide production, a rise in the vaginal pH, and the overgrowth of BV associated organisms.
(See Figure 1)
Gardnerella vaginalis is the most common bacteria associated with BV. Other less common ones are: Mycoplasma
hominis, Peptostreptococcus sp., Prevotella sp., Mobiluncus sp., Bacteroides sp. and Porphyromonas sp.
Transmission: BV is not currently considered to be a sexually transmitted infection but can be brought
on by anything that changes the balance in the vagina (e.g new partners, increased sexual activity).
Symptoms: Gardnerella is found in 50% of normal healthy women without causing any symptoms. Treatment
is not required in asymptomatic women unless they are about to undergo an invasive procedure such as IUCD insertion and
TOP.
For a quick diagnosis, some doctors test the pH of the vaginal discharge using litmus paper (normal vaginal pH is up
to 4.5), while others perform a “whiff” test which involves placing one drop of 10% potassium hydroxide (KOH)
onto a slide (onto which some discharge has been rubbed) and if BV is present it will cause the release of a fishy amine
odour.
BV often presents with a malodorous (fishy) grey/white vaginal discharge and only sometimes causes irritation and itching.
Testing: Swabs for BV are taken from the high vagina. The microbiological diagnosis is best made from
the gram stain appearance.
Non-specific urethritis - males only
Non-specific urethritis (NSU) or non-gonococcal urethritis (NGU) is a diagnosis of exclusion. It is defined as a condition
where the male urethra is inflamed (i.e there may be penile discharge, or numerous polymorphs on a urethral swab) but
a diagnosis of Chlamydia or Gonorrhoea has been excluded.
The prevalence figures for NSU in New Zealand have fluctuated over the past five years with the highest case numbers
seen in the age group 20-24 years.1
It is thought that some cases are caused by infection with organisms such as Mycoplasma genitalium, Ureaplasma urealyticum,
HSV and Trichomonas vaginalis or following exposure to irritants such as detergents or chemicals. Sometimes a cause is
never identified.
Genital warts

Background: Genital warts are the most common viral infection diagnosed in sexual health clinics, and
are most common in sexually active adolescents and young adults.
In the past five years there has been a 9.5% increase in people presenting to sexual health clinics in New Zealand with
genital warts.1
Approximately 90% of genital warts are caused by Human Papilloma virus (HPV) subtypes 6 & 11. They are of public health
concern because of their association with other subtypes of HPV (mainly 16 &18) that are linked with genital cancer (cervical,
penile and anal). The HPV vaccine Gardasil is now funded in New Zealand for females aged 12-18 years. For further information
see BPJ 18 (Dec 2008).
Transmission: HPV infection is transmitted by direct skin-to-skin contact and condoms are considered
only partially protective.
Symptoms: HPV infection can range from being asymptomatic to causing multiple fleshy or flat lumps
on the genitals. Lesions on the vaginal walls and cervix may be noted during PV examination.
Testing: At present, diagnosis is made clinically or the presence of HPV may be detected on smears.
Molluscum contagiosum
Molluscum contagiosum is caused by a pox virus. It causes a skin infection that appears as firm flesh-coloured bumps,
often with waxy white centres. Molluscum contagiosum is spread by close skin-to-skin contact, and occasionally via infected
clothing and towels. While it is frequently sexually transmitted, it is also common in children and people who are immunosuppressed.
Molluscum contagiosum is usually painless, although lumps may sometimes be itchy, or become infected. Lumps normally
appear about 2-3 months after infection, and without treatment they may persist for up to 2 years. It is recommended sexual
partners are advised to see their doctor, if they have any unusual genital lumps or itches.12
Donovanosis (Granuloma inguinale)
Donovanosis is an STI caused by Klebsiella granulomatosis. It can cause painless, progressive ulceration which sometimes
mimics genital cancer. Although endemic in some tropical areas, it is not currently considered a problem in New Zealand.
Diagnosis is by taking scrapings or collecting some tissue from the ulcers and looking for typical “Donovan bodies” seen
on staining.
Candidiasis (thrush)

Background: Genital thrush is usually caused by the yeast Candida albicans, though sometimes it can
be caused by atypical yeasts (such as C. glabrata) which can sometimes account for treatment failure. Women are more commonly
affected than men. About 10-20% of women in their reproductive years may be colonised with candida sp., but will be asymptomatic13 and
not require treatment.
Transmission: is not usually sexually transmitted and is most often associated with recent antibiotic
use, pregnancy, immunosuppression and diabetes. Many women also report thrush occurring more frequently while using oral
contraceptives, or at particular stages of their menstrual cycle.
Symptoms: can include itching/irritation around the vulva, vagina or foreskin, the presence of a white
discharge and dysuria.
The definition of recurrent thrush is four or more episodes/year, and this should trigger excluding conditions such
as diabetes or atypical yeast.
Testing: Swabs can be taken from the high vagina or male urethra/foreskin.
Specimen labelling requirements
Every specimen should have a MINIMUM of two patient identifiers:
- Patient family name and first name
- Patient date of birth and/or NHI number
Then ideally:
- The specimen site should be recorded on a swab label
- Date and time of specimen collection on request form and specimen label
Specimen collection
Laboratories in New Zealand can usually test for Gonorrhoea, Trichomonas vaginalis, Bacterial vaginosis and Candida
on the same swab. However, a separate Chlamydia swab is needed for Chlamydia, and a viral medium swab is needed for Herpes
Simplex Virus.
Specimen collection requirements may differ from region to region, check with your laboratory provider.
Partner notification
Partner notification is an essential component of STI management. It is worth remembering there are many ways of contacting
previous sexual partners. This may include email, texting, phone, face-to-face or letter.
| The current recommendations from the MOH Chlamydia guideline for partner notification are: |
- All sexual contacts within the previous 60 days need to be notified that they require testing and treatment for
possible chlamydia infection.
- If there have been no sexual contacts within the previous 60 days, then the most recent sexual contact should be
notified, up to a maximum of six months.
- All sexual contacts within the specified timeframe should be treated for chlamydia, even if the offer of a test
is declined.
|
- Patients should be offered the choice of:
- patient referral, where patients themselves notify their sexual contacts to seek treatment
- provider referral, where the health care provider agrees to undertake the task of notifying sexual contacts to
seek treatment (taking care to protect index case confidentiality as much as possible).
|
References
- Public Health Surveillance. The Institute of Environmental & Scientific Research (ESR) Annual report 2007. STI surveillance.
- Ministry of Heath. Chlamydia management guidelines, 2008. Available from
http://www.moh.govt.nz/moh.nsf/pagesmh/8210/$File/chlamydia-management-guidelines.pdf
- Cook R, Hutchison S, Ostergaard L, et al. Systematic review: noninvasive testing for Chlamydia trachomatis and Neiserria
gonorrhoeae. Ann Intern Med 2005;142(11):914-25.
- Schachter J, Chernesky M, Willis D et al. Vaginal swabs are the specimens of choice when screening for Chlamydia
trachomatis and Neisseria gonorrhoeae: results from a multicenter evaluation of the APTIMA assays for both infections.
Sex Transm Dis 2005;32(12):725-8.
- Wiessenfeld H, Lowry D, Heine R et al. Self-collection of vaginal swabs for the detection of Chlamydia, gonorrhea
and trichomonas: opportunity to encourage sexually transmitted disease testing among adolescents. Sex Transm Dis 2001;28(6):321-5.
- Chernesky M, Hook E, Martin D et al. Women find it easy and prefer to collect their own vaginal swab to diagnose
Chlamydia trachomatis or Neiserria gonorrhoeae infections. Sex Transm Dis 2005;32(12):729-33.
- Nieuwenhuis R, Ossewaarde J, Gotz H et al. Resurgence of Lymphogranuloma venerum in Western Europe: an outbreak of
Chlamydia trachomatis serovar L(2) proctitis in the Netherlands among men who have sex with men. Clin Infect Dis 2004;39(7):996-1003.
- Azariah S. Is syphilis resurgent in New Zealand in the 21st century? A case series of infectious syphilis presenting
to Auckland Sexual Health Service. NZ Med J 2005; Vol 118, No 1211.
- Cunningham R, MacDonald J, McLean M, Shaw C. An outbreak of infectious syphilis in Wellington, New Zealand. NZ Med
J 2007; Vol 120, No 1260.
- Beauman J. Genital herpes: a review. Am Fam Physician 2005;72(8):1527-34.
- Wilson J. Managing recurrent bacterial vaginosis: clinical knots. Sex Trans infect 2004;80:8-11.
- Auckland Sexual Health Service. The facts about Molluscum Contagiosum, 2007. Available from
http://www.sexfiles.co.nz/pdf_2007/molluscum.pdf
- British Association for Sexual Health and HIV. Guideline “Management of vulvocandidiasis” 2007 .Available
from: http://www.bashh.org/guidelines