Published: 19 January, 2024
Key practice points:
- Behavioural interventions and good sleep hygiene remain first-line for managing most forms of sleep disturbance. However, there can be instances where this approach is insufficient and pharmacological treatment needs to be considered.
- Melatonin is available in modified- or immediate-release formulations. While the rise in levels provided by either can potentially improve sleep parameters for some people, efficacy may be formulation- and indication-specific.
- In New Zealand, 2 mg modified-release melatonin tablets are approved but not funded for the short-term treatment of insomnia in adults aged 55 years or older. There is evidence that melatonin can reduce sleep onset latency and improve subjective measures of sleep quality in some people. This is the only approved form and use of melatonin.
- When used for this approved indication, 2 mg modified-release melatonin tablets can also be supplied by a registered pharmacist without a prescription
- Melatonin 2 mg modified-release tablets can be prescribed fully funded with Special Authority approval for the treatment of persistent and distressing insomnia secondary to a neurodevelopmental disorder in patients aged 18 years or under, where behavioural and environmental approaches have been tried and were unsuccessful or are inappropriate (unapproved indication)
- Initial Special Authority applications and renewals must be made by a psychiatrist, paediatrician, neurologist, respiratory specialist or on their recommendation
- Melatonin can also be considered for various other unapproved indications, including the treatment of insomnia in adults aged under 55 years, delayed sleep phase disorder, preventing or reducing jet lag and improving sleep in shift-workers. Data regarding efficacy in these situations is limited, with considerable inconsistency in clinical trial methodology and reported outcomes.
- Melatonin is generally well tolerated with short-term use. However, there is a lack of studies on potential long-term adverse effects, particularly in children and adolescents.
- If melatonin is being considered for any indication, discuss the importance of consistently taking it at the correct time each day (the modified-release formulation is generally taken one to two hours before bedtime); incorrect timing of use can result in daytime sleepiness or have other consequences on circadian rhythms
 This is a revision of a previously published article (BPJ 69, August 2015).
This is a revision of a previously published article (BPJ 69, August 2015).
What’s new for this update:
- General update of evidence
- Funding status of melatonin updated
- Focus on the role of melatonin in the management of primary insomnia
- Practice tips for unapproved uses of melatonin
Melatonin is a hormone that regulates circadian rhythm and sleep.1 It is synthesised from the amino acid tryptophan in the pineal gland and then released into the bloodstream and cerebrospinal fluid.1 This process is regulated by a “biological clock” in the suprachiasmatic nucleus (SCN) located within the hypothalamus and synchronised to the light/dark cycle.1
Total melatonin production levels change with age. Children begin synthesising and secreting melatonin from around age three months and total levels peak prior to puberty.1 Production then declines progressively and variably throughout adulthood; by the age of 80 years the nocturnal peak melatonin concentration can be less than one-third of young adult levels.2
Melatonin release is linked to circadian rhythm
Melatonin release from the pineal gland is suppressed by ocular light sensed at the retina.1 In a person with a normal sleep-wake cycle, melatonin is released at night, usually beginning 14 hours after spontaneous awakening, e.g. at 9 pm in a person who wakes at 7 am.1 Melatonin levels typically peak at approximately 2 – 3 am; secretion then decreases and, due to its relatively short biological half-life of 40 minutes, melatonin is undetectable in the bloodstream by approximately 6 – 7 am.1
Melatonin release, among other factors, therefore provides time-of-day information to numerous organs and tissues throughout the body, allowing certain physiological processes to align with the circadian cycle. The most extensively investigated roles of endogenous melatonin relate to control over sleep onset latency (i.e. the time it takes to fall asleep), total sleep time and overall sleep quality, but it also has a range of additional actions (see: “Endogenous melatonin does more than regulate sleep”).1
Factors that can disrupt the cycle of melatonin release:1,3–5
- Nocturnal light exposure. Artificial sources of light at night can have a suppressive effect on melatonin release; this effect is most notable with light emitted from digital screens, LED and fluorescent bulbs, and when it is of a blue wavelength.6 Even dim sources of light can have some influence on melatonin release, e.g. night lights. People who work night shifts have a delayed peak in melatonin secretion, the degree of which is influenced by the regularity and number of nights worked.
- Season and latitude-related differences. The duration of melatonin secretion is increased during longer winter nights (and reduced during the summer). This effect is particularly pronounced in people living at extreme northern or southern latitudes.
- Puberty. In addition to age-related changes in the quantity of melatonin produced, the rhythm of release is delayed as children enter puberty, resulting in later onset of sleepiness and a later natural wake time.
- Co-morbidities and medicine use. Certain conditions (e.g. neurodegenerative diseases, depression) and medicines (e.g. beta blockers, non-steroidal anti-inflammatory drugs) can influence melatonin production and release.
Given its diverse biological functions (see: “Endogenous melatonin does more than regulate sleep”), there is interest in the use of melatonin as a medicine. However, a key question is whether administering exogenous melatonin significantly influences the physiological processes it is associated with, many of which are also controlled by a network of additional factors.
The only validated indications for melatonin relate to sleep disturbances. Almost all people will experience dissatisfaction with sleep quality or distress from not sleeping at some point in their lives.7 Small reductions in sleep duration are also common as people age, even in the absence of sleep problems. In some people, sleep disturbances can significantly impact daily functioning and quality of life. Most evidence relating to the therapeutic use of melatonin involves treating people with insomnia as the relationship between melatonin and the sleep-wake cycle is well established (see: “The role of melatonin in the management of primary insomnia” and “Melatonin for insomnia in children with neurodevelopmental disorders”).8 The nightly melatonin peak is often altered (i.e. shifted or diminished) in those who report problems with the quality or quantity of their sleep.1 When given as a treatment for insomnia, melatonin is referred to as a chronobiotic, i.e. it alters the timing of sleep, as opposed to a hypnotic medicine (e.g. zopiclone, benzodiazepines) which act to rapidly initiate and prolong sleep.1, 8 There is also limited evidence that melatonin can be effective in treating other forms of sleep disturbance (see: “Other unapproved uses of melatonin”).
 Melatonin supplementation (or use of novel derivatives) has been considered in preliminary trials for the treatment or prevention of conditions involving oxidative stress, e.g. Parkinson’s disease, Alzheimer’s disease and cancer.1, 9 However, considerable further investigation is required, and there are no recommendations in current clinical guidelines for melatonin use in such contexts.
Melatonin supplementation (or use of novel derivatives) has been considered in preliminary trials for the treatment or prevention of conditions involving oxidative stress, e.g. Parkinson’s disease, Alzheimer’s disease and cancer.1, 9 However, considerable further investigation is required, and there are no recommendations in current clinical guidelines for melatonin use in such contexts.
Modified-release and immediate-release melatonin
Melatonin is available in modified- and immediate-release formulations.10 Modified-release melatonin causes plasma levels to more closely mimic a naturally occurring melatonin profile over time (Figure 1), whereas immediate-release melatonin results in a rapid increase in plasma levels and more significant peak.11, 12
Large head-to-head trials comparing modified- and immediate-release melatonin formulations are lacking. There is limited evidence suggesting the initial rise in melatonin levels provided by either is sufficient to reduce sleep onset latency,1, 11 but use of modified-release melatonin may additionally improve perceived sleep quality and morning alertness.11
Clinical response can, however, be unpredictable given that:1
- The timing, amplitude and other characteristics of a “normal” melatonin profile varies; and
- The capacity of a set dose of exogenous melatonin to influence this profile can be affected by individual variation in absorption, distribution, metabolism and elimination of melatonin, as well as the timing of intake and formulation selected
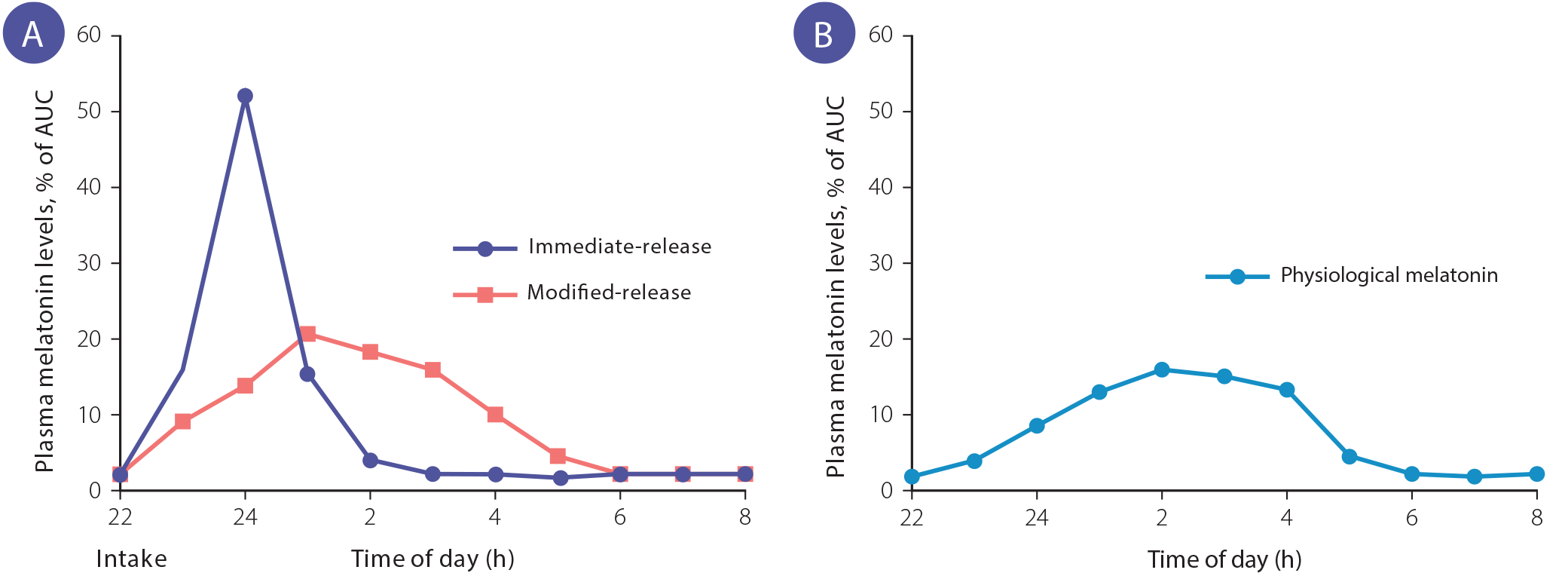
Figure 1. Mean plasma levels over time for (A) exogenous immediate-release and modified-release 2 mg melatonin formulations and (B) endogenous (physiological) melatonin. Plasma concentration levels are expressed as percentage of the area under the curve (AUC). Adapted from Zisapel, 2018.11
The status of melatonin in New Zealand
Melatonin is a restricted medicine in New Zealand. In some other countries, such as the United States, melatonin is considered a dietary supplement and can be purchased over the counter.
Melatonin 2 mg modified-release tablets are approved but not funded for the treatment of primary insomnia in adults aged 55 years or older, for up to 13 weeks.10 When used for this approved indication, 2 mg modified-release melatonin tablets can also be supplied by a registered pharmacist without a prescription.13 Modified-release melatonin is not approved in younger people (i.e. aged < 55 years) or for longer treatment durations (i.e. > 13 weeks) as clinical trials supporting efficacy and safety considered during the Medsafe approval process did not include these groups.
This is the only approved formulation and use of melatonin in New Zealand; all other uses are considered unapproved (“off-label”) and require a prescription (see: “Other unapproved uses of melatonin”).10, 13 Immediate-release melatonin is also available in New Zealand in various strengths but is not approved for any indication (Section 29). If in the future an immediate-release melatonin product is approved, there are provisions in a 2019 gazette notice for pharmacists to supply doses up to 3 mg for the treatment of primary insomnia in adults aged 55 years or older, for up to 13 weeks.
Using melatonin for the treatment of persistent and distressing insomnia secondary to a neurodevelopmental disorder in children and adolescents is also an unapproved indication, but is funded with Special Authority approval (see: “Melatonin for insomnia in children with neurodevelopmental disorders”).
Patients diagnosed with primary insomnia require an individualised approach to management. Unrealistic sleep expectations and poor sleep hygiene underpin insomnia in many patients, therefore the first-line treatment should be non-pharmacological interventions addressing these factors, including sleep hygiene education, stimulus control, bedtime/sleep restriction, relaxation techniques and cognitive behavioural therapy (CBT).14 Programmes relating to these principles are available online or as apps.
Short-term medicine use is sometimes considered as a supplement to behavioural interventions for patients who are not responding to CBT alone, or in those with severe symptoms at baseline.14 Options include zopiclone, benzodiazepines, tricyclic antidepressants or melatonin.10, 14 There is little evidence to guide the initial selection or order of trial prescribing in this context; choice between the different options is typically made based on patient risk factors and adverse effects. The British Association for Psychopharmacology (BAP) recommends that modified-release melatonin should be trialled before a hypnotic for the treatment of insomnia in adults aged 55 years or older.15 However, there are weak recommendations against using melatonin for the treatment of insomnia in American and European guidelines.16, 17
N.B. The distinction between “primary” and “secondary” insomnia has been removed in recent diagnostic classification systems (e.g. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition [DSM-5], International Classification of Sleep Disorders-3), however, these individual terms are still widely used in the literature.
For further information on the diagnosis and management of insomnia in adults see: “I dream of sleep: managing insomnia in adults”
A “living” resource on managing insomnia in primary care was developed in 2022 by the Australian National Centre for Sleep Health Services Research, with ongoing maintenance and updates provided by the Australasian Sleep Association. See: www.sleepprimarycareresources.org.au/insomnia
How much improvement in sleep can adults with primary insomnia expect when using melatonin?
Numerous trials have investigated melatonin for the treatment of insomnia in adults. However, evidence regarding its efficacy remains contentious; some studies report improvements in sleep efficiency and quality, whereas others report no differences between melatonin and placebo groups.18 Data interpretation is limited by differences in study cohorts, methodology and the outcomes investigated.
Of the studies that do report benefits for melatonin in adults with primary insomnia, effects are generally variable and modest. Those specifically focusing on people aged at least 55 years suggest that 2 mg modified-release melatonin can improve sleep onset latency by approximately ten minutes.19 Subjective improvements in sleep quality and morning alertness have also been demonstrated in older adults.19
Across all adult age groups, potential benefits are less compelling, with quantitative analysis of randomised controlled trials (RCTs) suggesting melatonin reduces sleep onset latency by approximately five minutes compared with placebo.19 Reported effects on total sleep duration with melatonin use in adults are inconsistent between studies;19 if present, any improvement is likely small.
Healthy children and adolescents often do not get enough sleep – melatonin is rarely the answer
Approximately 20% of children and adolescents experience problematic amounts of sleep-related difficulties; this is excluding children with neurodevelopmental disorders who experience higher rates of sleep disturbance (see: “Melatonin for insomnia in children with neurodevelopmental disorders”).20 For children, sleep difficulties usually manifest as bedtime resistance and sleep anxiety, night waking and parasomnias, whereas adolescents more commonly exhibit daytime sleepiness.20
In general, bedtimes become later and sleep duration reduces with increasing age due to changes in lifestyle and the timing of melatonin release.20 Teenagers also often have a more variable sleep pattern across the week as they go into sleep deficit during weekdays, while attending school and extracurricular activities, before recovering this debt over the weekend by sleeping longer. These “expected” changes in sleeping patterns make it difficult to distinguish the true prevalence of more serious sleep disorders in teenagers, e.g. insomnia.
Optimisation of bedtime behaviour and routines should be emphasised as the first step for sleep problems in most young patients. For example, interventions simply focusing on earlier bedtimes have been shown to increase total sleep time by almost 50 minutes in healthy children.21
On balance, melatonin use in otherwise healthy children and adolescents should generally be reserved for resistant cases where behavioural interventions and attempts at good sleep hygiene have not been successful, and daytime functioning is compromised (unapproved indication).22 Low quality evidence from an analysis of eight RCTs (N = 419) suggests melatonin supplementation increases total sleep time by 30 minutes and reduces sleep onset latency by 18 minutes in children and adolescents diagnosed with idiopathic chronic insomnia, in whom sleep hygiene practices have already been trialled and were inadequate.22 Despite these possible quantitative benefits, little to no effect on sleep quality or daytime functioning has been identified, and the risk of non-serious adverse effects is increased three-fold, e.g. headache, nausea.22 One study reported a potential increase in nocturnal awakenings, however, further investigation is required to corroborate this effect in healthy children.22
For further information on managing sleep problems in children, see:
General dosing principles of melatonin for insomnia
The timing and consistency of melatonin administration is important, as it may cause circadian phase advancement when taken early, or phase delay when taken late.23
Modified-release melatonin is usually taken one to two hours before intended sleep onset, with food or just after eating.10 Alcohol should be avoided after taking the dose as it can increase the speed of melatonin release into the blood stream, thereby effectively turning the modified-release formulation into an immediate-release medicine.10 Immediate-release melatonin is generally taken at bedtime. Patients should also be advised not to drive or operate machinery for the rest of the evening once they have taken either form of melatonin.
Dividing, chewing or crushing modified-release tablets is not recommended by the manufacturer – tablets should usually be swallowed whole.10 However, if tablets are broken for administration, patients should be advised that this will alter the characteristics of the medicine and confer immediate-release effects, which means it will need to be taken at bedtime.
Clinicians can request on the prescription that the pharmacy compounds melatonin into an oral liquid suspension (making it immediate-release), which may be useful in patients with swallowing difficulties (or in children who cannot swallow tablets).10
N.B. There is evidence that careful halving of Circadin-branded 2 mg modified-release tablets largely retains the modified-release profile.24 However, this characteristic has not been investigated or established for other brands of modified-release formulations.
Stopping melatonin
Melatonin is only approved for a treatment duration of 13 weeks when prescribed to adults aged ≥ 55 years with insomnia. Continued treatment beyond this point is considered unapproved use. However, in practice, melatonin is often continued longer-term, if it is providing benefit, there are no adverse effects and no other treatments are effective.
In most cases, melatonin can be stopped abruptly without needing to taper the dose; it is not associated with discontinuation effects.25, 26 There is no evidence that melatonin causes tolerance, dependence, withdrawal or rebound insomnia.26, 27
Melatonin for insomnia in children with neurodevelopmental disorders
Sleep disturbances are common in children with neurodevelopmental disorders, e.g. autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD), visual impairment, epilepsy, cerebral palsy.28 These can be challenging to treat, often persist into adolescence and have a significant impact on the wellbeing of the patient and their family/whānau.28 Specialist input is generally required to guide management.
In New Zealand, melatonin is dispensed to approximately 19% of children with ASD, 22% with ADHD and 30% diagnosed with both conditions (compared with 0.5% of children without these conditions).29
 Prescribing. Melatonin 2 mg modified-release tablets can be prescribed fully funded with Special Authority approval for the treatment of persistent and distressing insomnia secondary to a neurodevelopmental disorder in patients aged 18 years or under, where behavioural and environmental approaches have been trialled or are inappropriate and the dose does not exceed 10 mg per day (unapproved indication).30 Initial Special Authority applications and renewals must be made by a psychiatrist, paediatrician, neurologist, respiratory specialist or on their recommendation.30 In some cases, general practitioners may manage continued treatment under a shared-care agreement; the need to continue treatment should be reviewed every six months.30
Prescribing. Melatonin 2 mg modified-release tablets can be prescribed fully funded with Special Authority approval for the treatment of persistent and distressing insomnia secondary to a neurodevelopmental disorder in patients aged 18 years or under, where behavioural and environmental approaches have been trialled or are inappropriate and the dose does not exceed 10 mg per day (unapproved indication).30 Initial Special Authority applications and renewals must be made by a psychiatrist, paediatrician, neurologist, respiratory specialist or on their recommendation.30 In some cases, general practitioners may manage continued treatment under a shared-care agreement; the need to continue treatment should be reviewed every six months.30
 Dosing. The recommended dose in children aged one month to 18 years is 2 – 3 mg daily, before bedtime.30 This can be increased to 4 – 6 mg after one to two weeks, to a maximum of 10 mg daily (higher doses within this range should be used with caution). Modified-release melatonin tablets should be given one to two hours before bedtime (or at bedtime if administered in a compounded oral suspension).
Dosing. The recommended dose in children aged one month to 18 years is 2 – 3 mg daily, before bedtime.30 This can be increased to 4 – 6 mg after one to two weeks, to a maximum of 10 mg daily (higher doses within this range should be used with caution). Modified-release melatonin tablets should be given one to two hours before bedtime (or at bedtime if administered in a compounded oral suspension).
A closer look at the evidence
An analysis of 13 RCTs including 682 participants aged < 18 years diagnosed with both neurodevelopmental and sleep disorders demonstrated that melatonin is moderately effective at improving sleep. Total sleep time increased by 48 minutes with melatonin compared with placebo, and sleep onset latency was also improved (mean difference = 29 minutes).31 Melatonin use did not influence the frequency of nocturnal awakenings.31
A double-blinded RCT specifically investigated 2 mg modified-release melatonin for insomnia in 125 children with ASD (with or without ADHD). It was found that 13 weeks of treatment resulted in significantly more participants achieving a clinically meaningful response in total sleep time and/or sleep onset latency versus placebo (68.7% versus 39.3%; number needed to treat = 3.4).32 These benefits were sustained in a one-year follow-up report, which included participants receiving doses up to 10 mg.33 Continued treatment was generally well tolerated.33

Melatonin is generally very well tolerated with short-term use.26 Following initiation, possible adverse effects are diverse but relatively infrequent; if they do occur, most are mild to moderate in severity, and resolve spontaneously within a few days (or immediately once treatment is stopped).26 Rates of adverse effects are comparable between melatonin and placebo in RCTs, with the most common examples including daytime sleepiness, nausea, headache, other sleep-related adverse effects (e.g. vivid dreams), dizziness, asthenia (weakness) and musculoskeletal pain.26, 34 The comparative incidence and severity of daytime sleepiness associated with melatonin is lower than with hypnotic medicines such as zopiclone or benzodiazepines.26, 27 There has been no reported increased risk of cognitive impairment or falls, fractures or motor vehicle accidents associated with melatonin use in older patients.26
Interactions with other medicines
Melatonin is primarily metabolised by CYP1A enzymes in the liver, meaning it should be avoided in patients with hepatic impairment due to reduced clearance.10, 26 Interactions are possible when melatonin is taken concurrently with other medicines metabolised by CYP1A enzymes, however, in practice these effects are infrequently reported and not robustly quantified in the literature.1, 26 Medicines that may increase melatonin plasma concentrations include citalopram, cimetidine and quinolones (e.g. ciprofloxacin).10, 26 Carbamazepine and rifampicin may cause plasma concentrations of melatonin to be reduced.10
Practice point: Children may have reduced CYP1A2 activity compared to adults,35 which in addition to the above potential medicine interactions, may theoretically expose them to higher melatonin concentrations than anticipated.
More studies on the long-term safety of melatonin are needed
As of 2023, systematic reviews of both RCTs and observational studies have not confirmed an association between chronic melatonin use and long-term adverse effects in any age group.34, 36 Previously, there had been concerns that chronic use of melatonin may have unforeseen consequences, particularly among children and adolescents. For example, given that melatonin levels usually decline gradually before puberty in normal development, there was worry that supplementation may cause a delay in puberty onset.34, 36 However, evidence suggests that two to four years of melatonin treatment has little to no influence on the timing of puberty,34 with one small survey-based study suggesting only a weak trend towards a delay in “perceived pubertal development” in participants after an average melatonin treatment duration of 7.1 years (N = 33).37 Another concern was that melatonin supplementation may cause adverse bone health outcomes due to its role in bone mineralisation during pubertal development, however, no studies to date have confirmed this link.34, 36
Nevertheless, given that only a limited number of trials have investigated the safety of long-term melatonin treatment (including a scarcity of robust double-blinded RCTs), caution is still recommended if longer periods of use are being considered.34, 36 Ideally, further research is required, including analysis of existing large health database records.
Patients may enquire about the suitability of melatonin treatment for indications other than primary insomnia, e.g. preventing jet lag or managing sleep in shift-work. In these situations, there may be few practical treatment options available and therefore those without a robust evidence base need to be considered. Table 1 provides an overview of evidence for unapproved uses of melatonin.
Patients prescribed melatonin for off-label purposes should be told that there is very limited and inconsistent data available concerning the efficacy or safety of the medicine for this use, and that the details of the prescription will be recorded on a database as a requirement of the Medicines Act.38 In many cases of off-label use, there is no established dosing regimen for melatonin; an appropriate protocol may be derived from clinical studies. Clinicians may prefer prescribing approved modified-release melatonin, but in some situations, evidence may suggest immediate-release melatonin is more effective. Discussing the patient with a sleep specialist may be beneficial before prescribing melatonin for an unapproved indication, although this will not always be practical.

Reminders if considering unapproved melatonin use1
- More does not always mean better. There is minimal evidence for additional benefit with melatonin doses > 5 mg daily in clinical trials; lower doses often have comparable efficacy to higher doses,39 but are less likely to cause adverse effects, e.g. excessive sedation before the desired bedtime. Starting at a lower dose and titrating upwards, if required, is therefore usually the best strategy.
- The timing of administration is important. The ideal timing of melatonin administration differs depending on the formulation used (i.e. modified- vs. immediate-release) and the desired effect (e.g. phase advancement, delay or no phase shift).40 Table 1 includes evidence related to dose timing for different unapproved indications.
- Check the ingredients. Quality control and assurance can only be provided for melatonin products with Medsafe approval. Unapproved melatonin products obtained from overseas suppliers may contain other ingredients that influence sleep (e.g. valerian, magnesium) and sometimes may even include prescription medicines. If sleep improves when using these combination products, it may be unclear which ingredient is responsible for the beneficial effect. Additional ingredients may also be associated with adverse effects or medicine interactions.
Table 1. An overview of evidence for unapproved (off-label) melatonin use.
N.B. This table does not include evidence relating to the management of persistent insomnia in children secondary to a neurodevelopmental disorder, which is also an unapproved indication but funded with Special Authority approval (for further information see main text).
| Off-label use |
Source of evidence |
Evidence overview |
Delayed sleep phase disorder19
- Encompasses people whose sleep and waking times are consistently later or mismatched with conventional or desired expectations, e.g. by greater than two hours (colloquially referred to as “night owls”)
- More common in adolescents/young adults
- Associated with ADHD and higher rates of smoking, alcohol use, caffeine use, depression
|
- Systematic review and meta-analysis including 34 studies, seven of which focused on melatonin for delayed sleep phase disorder41
|
- Melatonin may improve total sleep time in adults (by over 40 minutes) and sleep onset latency in children/adolescents (by over 25 minutes) versus placebo. However, individual responses vary significantly and depend on the reporting method (e.g. sleep study, sleep diary or wearable device).
- While both formulations have potential efficacy, most available evidence relates to immediate-release melatonin
- There is no consensus on the ideal dose or timing of administration in adults. Most study doses ranged from 0.5 – 5 mg, taken 30 minutes to two hours before the desired bedtime (sometimes even earlier). Usual treatment duration was four weeks.
- The NZFC recommends using oral immediate-release capsules or tablets, or oral suspension for children with delayed sleep phase disorder, initially 2 – 3 mg, daily, immediately before bedtime, increased if necessary after 1 – 2 weeks to 4 – 6 mg, daily, before bedtime. Maximum 10 mg daily.
|
Shift-work sleep disorder42
- Shift-workers often experience sleep disturbances resulting in sleepiness and impaired cognition when working at night, as well as reduced sleep duration/quality during the day
- In patients with extreme symptoms this is termed shift-work sleep disorder
|
- Cochrane systematic review including 15 RCTs, nine of which assessed the effects of melatonin in shift-workers42
- Systematic review including ten studies specifically focusing on melatonin for healthcare shift-workers43
|
- Melatonin may improve total sleep time during the day (by almost 25 minutes) and sleep length the next night after finishing shift-work (by over 15 minutes) versus placebo. There was inconsistent evidence for an effect on sleep onset latency between reviews. Evidence considered to be low quality.
- Melatonin was administered in the morning in most trials, after the night shift had finished, and before the daytime sleep period
- Formulation type was not reported. It was suggested that immediate-release effects would be more desirable in this context.
- Study doses ranged from 1 to 10 mg. No additional benefit was found at strengths above 5 mg. A low initial dose (e.g. ≤ 2 mg) was noted as being suitable if melatonin is being trialled (i.e. to establish any clinical effect).
|
Sleep disturbance in adults with complete vision loss (blindness)44
- Sleep disturbances are much more common in people with blindness versus the general population
- Impaired transmission of light between the retina and circadian clock in the SCN leads to desynchronisation with external day/night cycle (also known as non-24 hour sleep-wake disorder)
- Results in chronic or cyclical episodes of poor sleep and impaired daytime functioning
|
- Narrative review including five studies44
- NICE evidence summary of three studies45
|
- Only three trials directly reported on the clinical effect of melatonin for sleep (one RCT, two crossover studies [uncertain randomisation]). These included very small sample sizes (7 – 13 patients).
- The only RCT (N = 13) found that six weeks of treatment with 2 mg modified-release melatonin reduced sleep onset latency almost 30 minutes and increased total sleep time by over 25 minutes versus placebo. Evidence reported as not being statistically significant but potentially clinically significant.
|
Jet lag46, 47
- Can occur when travelling across at least two time zones
- Involves circadian rhythm becoming desynchronised with the day-night cycle of travel destination
- Characterised by daytime fatigue and sleep disturbance; may also include reduced cognitive function, dizziness, weakness and irritability
- May take up to four to six days for synchronisation to occur in travel destination
|
- Cochrane systematic review including ten studies on melatonin for jet lag.46 N.B. Published in 2002.
- Narrative review on general pharmacological management of jet lag, including more recent melatonin studies47
|
- Melatonin use potentially reduces impact or duration of symptoms*
- Effect is most significant in people travelling across five or more time zones in an eastward direction (e.g. Auckland to Chicago) as they require circadian advancement for readjustment
- Most evidence relates to immediate-release melatonin; one small head-to-head trial found it more effective than the modified-release formulation
- Ideal dose and timing are uncertain. Doses between 0.5 – 5 mg have a largely similar efficacy. With eastward travel, take melatonin before bedtime after arrival in travel destination. Ideal timing for westward travel likely differs but there are few trials to establish clear guidance.
- Doses may be repeated for several days after arrival (taken at same time)
- N.B. Do not use melatonin prior to departure at a time corresponding to bedtime at the destination (i.e. to anticipate the change in time zone).
|
*Timed exposure to bright light (preferably sunlight or artificial full spectrum light) can also assist in adjusting to a new time zone.43 However, as with melatonin use, efficacy is influenced by the direction of travel and the degree of desynchronisation.43 For example recommendations, see: www.caa.co.uk/Passengers/Before-you-fly/Am-I-fit-to-fly/Health-information-for-passengers/Jet-lag
ADHD = attention deficit hyperactivity disorder; NICE = National Institute for Health and Care Excellence; NZFC = New Zealand Formulary for Children; RCT = randomised controlled trial; SCN = suprachiasmatic nucleus
Endogenous melatonin does more than regulate sleep
Melatonin receptors are broadly distributed throughout the body; both within the central nervous system (CNS) and in peripheral tissues and organs.1 As such, endogenous melatonin is reported to have a range of actions in addition to regulation of sleep. However, there is no compelling evidence that supplementation with exogenous melatonin has a significant clinical effect on these additional processes.
Physiological processes influenced by endogenous melatonin (to variable extents) include:
- Thermoregulation – peak melatonin levels are associated with troughs in the circadian rhythm of body temperature1
- Blood pressure – melatonin secretion at night reduces blood pressure.1, 48 Proposed mechanisms include endothelium-dependent influences on vasodilation and changes to sympathovagal autonomic regulation. Melatonin also has other effects on the cardiovascular system, e.g. heart rate control, cardioprotection.49
- Neural function – melatonin promotes a range of neural functions within the brain and CNS, including neurogenesis, neurotransmission and neuroprotection1, 50
- Immune response – melatonin is involved in regulating aspects of both innate and adaptive immunity (including anti-inflammatory actions)1, 50
- Energy metabolism and glucose homeostasis – evidence from animal studies suggests a possible role for melatonin in regulating glucose levels and cellular metabolism (in addition to other key regulators, e.g. insulin).50, 51 The nature and extent of this relationship in humans is a subject of ongoing investigation. Melatonin receptors are expressed within adipose tissue and may influence fat accumulation.
- Reproductive function – melatonin can influence the timing of sexual/developmental maturation and reproductive function, e.g. by inhibiting gonadotropin-releasing hormone expression1, 50
- Antioxidant properties (see below)1, 50
Melatonin is a powerful antioxidant
Melatonin is an “ancient” molecule in an evolutionary sense and is present in a diverse range of organisms, including vertebrates, insects, plants, cyanobacteria and fungi.1 While its receptor-mediated roles vary between organisms, one conserved function is melatonin’s antioxidant properties, allowing it to act as a free radical scavenger.1, 50 Melatonin readily penetrates cellular membranes and can provide antioxidant protection to mitochondria and DNA.1 There is in vitro and in vivo evidence indicating melatonin may have a protective effect against the development of cancer, both by eliminating free radicals produced by UV radiation (e.g. reactive oxygen species) and promoting DNA repair.1, 9 However, no meaningful clinical effects have been observed in humans for this purpose.





