Key practice points:
- The typical presentation of polymyalgia rheumatica (PMR) is a recent onset of aching pain and morning stiffness lasting at least 30 – 45 minutes, affecting both shoulders and often the neck, the hip girdle and occasionally the lower back. It is almost exclusively seen in people aged > 50 years of European ethnicity.
- A diagnosis of PMR is made clinically based on recognition of a history of characteristic symptoms, with a raised CRP (or ESR if CRP is normal), exclusion of other conditions which may mimic the history (e.g. rheumatoid arthritis) and a rapid response to oral corticosteroid treatment
- Giant cell arteritis (GCA), a form of vasculitis, has a strong association with PMR and up to one-fifth of people with PMR will develop GCA at some point. Therefore, a high suspicion for GCA is needed before and during treatment for PMR to prevent ischaemic complications, e.g. permanent blindness.
- Symptoms or signs of GCA include temporal artery or scalp tenderness, new onset or type of headache, jaw claudication, visual disturbances. If GCA is suspected, refer the patient for an urgent temporal artery biopsy or discuss with the rheumatology service according to local protocols. Initiate high dose oral corticosteroids as soon as the diagnosis is suspected.
- Assessment of the patient’s response to treatment for PMR (usually prednisone) should primarily be based on their clinical response, e.g. ability to perform movements and tasks that were previously impaired (such as getting out of a chair). Symptoms generally improve rapidly after treatment initiation, i.e. within a few days to one week.
- An inadequate response to prednisone within seven to ten days is uncommon and if this occurs, the diagnosis should be reconsidered. If clinical suspicion for PMR remains, consider trialling a higher prednisone dose after discussing with the local rheumatology service.
- Monitoring CRP (or ESR) levels is often helpful when assessing a patient’s response to prednisone; these should normalise within two to four weeks (however inflammatory marker levels may not always correlate with the patient’s symptomatic response)
- Once there is adequate improvement in the patient's symptoms, attempt to slowly and progressively taper the corticosteroid dose; the rate of taper should be done primarily in response to the patient’s symptoms, not CRP (or ESR) results alone
- Relapses are not uncommon during this time; if they occur, increase the dose back to pre-relapse levels
- PMR is usually a self-limiting condition, and does not appear to cause a risk of premature mortality
- While the aim is to stop corticosteroid use as soon as possible, the average duration of treatment is one to two years
- Long-term treatment decisions are individualised, taking into consideration the adverse effects associated with prolonged corticosteroid use, e.g. osteoporosis, diabetes, gastritis, adrenal insufficiency
- For patients who require an alternative to corticosteroids, treatment decisions should be made following consultation with a rheumatologist; methotrexate is an option
- This article features audio commentary from Professor Simon Stebbings, Consultant Rheumatologist and Head of the Rheumatology Research Unit, Dunedin School of Medicine, University of Otago.
This is a revision of a previously published article. What’s new for this update:
- A general article revision
- Audio commentary featuring expert Professor Simon Stebbings
- Updated corticosteroid dose tapering regimen
- Section added on giant cell arteritis
Polymyalgia rheumatica (PMR) is an inflammatory rheumatological condition characterised by a distinctive pattern of joint pain and morning stiffness (see: “Typical presentation of PMR”).1, 2 It is the second most prevalent autoimmune rheumatic condition in adults after rheumatoid arthritis, and it is the most common rheumatic condition among people aged > 50 years.3
- The incidence of PMR increases with age; almost all people affected are aged > 50 years, with peak incidence between the ages of 70 – 75 years.2, 4
- PMR is two to three times more common in females than males.5, 6 The lifetime risk of PMR has been reported as 2.4% in females and 1.7% in males.7
- Most people affected by PMR are of European ethnicity.5 Incidence varies geographically but is highest in people of Northern European ethnicity, particularly those from Scandinavian countries.5, 8 PMR is less common in Southern European countries and is particularly uncommon in African and Asian populations, however, it can affect people of any ethnicity.5 Data are unavailable in New Zealand, but anecdotally PMR rarely occurs in people who are non-European.
The cause of PMR remains ambiguous.5 The inflammatory processes predominantly affect synovial and periarticular structures (bursae and tendons) rather than muscles.5, 8 FDG-PET/CT scans have identified the most common sites of inflammation to be the shoulder and hip joints, sternoclavicular joints and lumbar interspinous processes.9 The immunopathology of PMR is currently understood to be due to both autoinflammation and autoimmunity, the former process being driven by an ageing immune system.4 Unlike many other rheumatic or inflammatory conditions, autoantibodies are not a feature in people with PMR.5, 8 It is thought that genetic susceptibility contributes to geographical variation, e.g. people from Scandinavian countries have a high prevalence of PMR, while people from Finland, who are a genetically different population, have a much lower prevalence.5, 10 Seasonal differences may be due to an infectious trigger, e.g. virus.5, 10 It has been suggested that hormonal influences on autoimmunity may explain differences in the prevalence of PMR between sexes, but there is conflicting information on this.
There is no specific test or other investigation to definitively diagnose PMR, and symptoms and signs can overlap with other conditions that cause inflammation and joint pain.5, 11
In practice, a diagnosis of PMR is based on recognition of a history of characteristic symptoms, with a raised C-reactive protein (CRP; or erythrocyte sedimentation rate [ESR] if CRP is normal), exclusion of other conditions which may mimic the history (e.g. rheumatoid arthritis) and a rapid response to oral corticosteroid treatment (Figure 1).5 The treatment of patients with PMR requires long-term oral corticosteroids which can be associated with significant adverse effects, so diagnostic certainty is important.
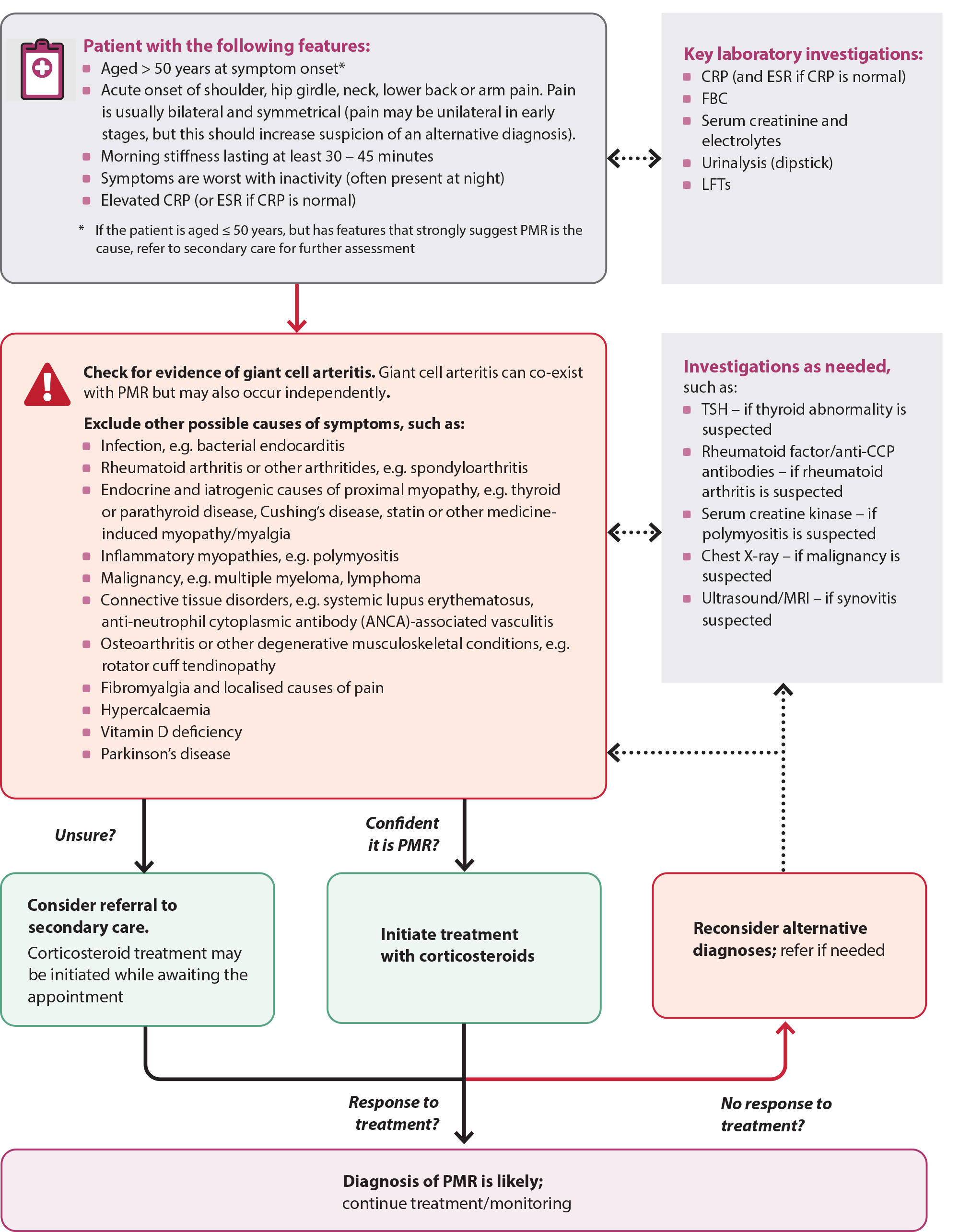
Figure 1. Diagnostic overview of polymyalgia rheumatica.3–6, 8, 12
CRP = C-reactive protein; ESR = erythrocyte sedimentation rate; FBC = full blood count; LFTs = liver function tests; TSH = thyroid stimulating hormone; anti-CCP = anti-cyclic citrullinated peptide; PMR = polymyalgia rheumatica
Typical presentation of PMR
The onset of PMR is usually rapid, with symptoms developing overnight or within a few days.1, 5 For some people onset can be more insidious, occurring over several weeks to months, however, a longer symptom onset should raise suspicion of an alternative diagnosis.1, 5
Shoulder, hip, neck, lower back and arm pain. Shoulder pain occurs in almost all people with PMR.8, 13 Hip girdle and neck pain is also common, occurring in more than half of cases.8 Pain can also be present in the upper arms and lower back, and can radiate to the elbows and knees in some cases.4, 8 Pain is usually bilateral and symmetrical, although it may be worse on one side early in the course of the condition.4, 13 Night pain can be typical and some patients may describe sleep disturbances.8, 13
There may be tenderness on examination, which is usually related to synovial or bursal inflammation.1, 13 Muscle weakness is not a feature of PMR, although this can be difficult to assess due to myalgia.8, 13
Stiffness. Marked morning stiffness that persists for at least 30 – 45 minutes is a typical feature of PMR.1, 4, 8 Patients may describe difficulties with daily activities, such as turning over in or getting out of bed, getting dressed, brushing their hair and rising from a chair.4, 13 Stiffness and pain typically lessen over the course of the day but may be worse after rest or with inactivity.8, 13
Peripheral symptoms and signs. Pain or stiffness in distal joints can be present in approximately half of people with PMR; examine the hands, feet, knees and elbows for signs of joint inflammation.8, 14 Peripheral signs such as joint swelling are common in other similar conditions and may indicate an alternative diagnosis such as rheumatoid arthritis or other inflammatory arthritides.3, 4
Systemic symptoms. Up to half of all people with PMR experience systemic symptoms such as a low-grade fever, fatigue, loss of appetite, malaise and weight loss.12, 13 People aged > 70 years are more likely to experience systemic symptoms.4, 14
 Always consider GCA. GCA must be treated urgently to prevent ischaemic complications, e.g. permanent blindness.13, 15 Ask about symptoms that may suggest GCA such as new onset or type of headache, temporal artery or scalp tenderness, jaw claudication and visual symptoms.5, 13 If GCA is suspected, refer the patient for an urgent temporal artery biopsy (or discuss with the rheumatology service according to local protocols) and initiate high dose corticosteroids.15, 16 For further information, see: “Are cranial symptoms present? The patient may have giant cell arteritis”.
Always consider GCA. GCA must be treated urgently to prevent ischaemic complications, e.g. permanent blindness.13, 15 Ask about symptoms that may suggest GCA such as new onset or type of headache, temporal artery or scalp tenderness, jaw claudication and visual symptoms.5, 13 If GCA is suspected, refer the patient for an urgent temporal artery biopsy (or discuss with the rheumatology service according to local protocols) and initiate high dose corticosteroids.15, 16 For further information, see: “Are cranial symptoms present? The patient may have giant cell arteritis”.
Initiating further investigations
If the patient’s history and examination suggests that PMR is likely, the following tests should be requested:1, 4
- CRP (and ESR if CRP is normal)
- Full blood count (normochromic normocytic anaemia, thrombocytosis and leukocytosis may be present)
- Serum creatinine and electrolytes
- Urinalysis (dipstick)
- Liver function tests (approximately one-third to half of people with PMR have mildly elevated liver enzymes, particularly alkaline phosphatase)
There is no gold standard test for the diagnosis of PMR, but elevated acute phase response markers such as CRP and/or ESR should increase suspicion as these are typically raised in a patient with PMR.5, 13 Normal inflammatory markers although rare (likely under 1%), cannot exclude PMR as a cause of the patient’s symptoms, and further investigation is required.5, 13 If CRP is > 100 mg/L, consider referral for a temporal artery biopsy, even if the patient does not have classical GCA symptoms.
 Listen to Simon Stebbings discuss use of inflammatory markers, particularly CRP, for the diagnosis of PMR
Listen to Simon Stebbings discuss use of inflammatory markers, particularly CRP, for the diagnosis of PMR
Depending on the patient’s symptoms and signs, additional laboratory tests may be requested to rule out other potential diagnoses, including:2, 14
- Thyroid stimulating hormone – if thyroid abnormality is suspected
- Rheumatoid factor/anti-cyclic citrullinated peptide antibodies – if rheumatoid arthritis is suspected
- Serum protein electrophoresis (consider serum free light chain assay if electrophoresis is negative) – if a myeloproliferative disorder is suspected
- Serum creatine kinase – if polymyositis is suspected
- Antinuclear antibodies – if autoimmune or connective tissue disorder is suspected, e.g. systemic lupus erythematosus
Imaging is not required for the diagnosis of PMR
Imaging modalities are not usually required for the diagnosis of PMR as findings are often unremarkable, however, they may be used in select patients where a diagnosis of PMR is uncertain.2, 5
- X-rays of affected joints will usually be normal and therefore are not required for investigating PMR (chest X-ray may be required if malignancy is suspected).1
- If ultrasound is accessible, assessment of the shoulder and hip joints can be considered.5, 13 Bursitis and synovitis are common findings in people with PMR, but are non-specific.5, 13
Additional imaging such as CT or MRI and very occasionally FDG-PET/CT, may be used in a secondary care setting to identify bursitis, synovitis or tenosynovitis in the shoulder and hips, and for excluding other potential diagnoses.2, 5, 8
Are cranial symptoms present? The patient may have giant cell arteritis
Giant cell arteritis (GCA), a form of vasculitis, involves inflammation of the blood vessels of the head, neck and chest. This condition is closely related to PMR (Figure 2), however, the relationship is not completely understood.10, 15 GCA can also occur in the absence of PMR.
People with PMR
- PMR is at least three times more prevalent than GCA
- Up to 21% of people with PMR have co-existing GCA
- 40 – 60% of people with GCA have concurrent PMR
- Like with PMR, GCA is most common in European females aged > 50 years
Figure 2. The relationship between GCA and PMR.10, 15
 Symptoms or signs of GCA can include:10, 12, 15
Symptoms or signs of GCA can include:10, 12, 15
- New onset or new type of headache
- Temporal artery or scalp tenderness
- Jaw or tongue claudication
- New and unexplained visual impairment, e.g. diplopia, reduced visual acuity, acute visual loss
- Fever, anorexia
- Unexplained elevated levels of CRP +/- ESR
 If GCA is suspected, request a temporal artery biopsy (or discuss with the rheumatology service according to local protocols*) and initiate treatment with high dose oral corticosteroids (see dosing below) as soon as possible to prevent ischaemic complications, e.g. permanent visual impairment, aortic dissection or aneurysm, transient ischaemic attack/stroke.15, 16
If GCA is suspected, request a temporal artery biopsy (or discuss with the rheumatology service according to local protocols*) and initiate treatment with high dose oral corticosteroids (see dosing below) as soon as possible to prevent ischaemic complications, e.g. permanent visual impairment, aortic dissection or aneurysm, transient ischaemic attack/stroke.15, 16
*Some rheumatology services are currently implementing temporal artery ultrasound into the diagnostic algorithm for GCA
Do not wait for the results of the biopsy before initiating treatment.12 Where there is a strong clinical suspicion of GCA, a delay in treatment will almost always have greater consequences than an unnecessary dose of corticosteroids in a patient who is later found to not have GCA (but the patient may still have PMR). The risks of potentially over-treating uncomplicated PMR are greatly outweighed by the risks of undertreating GCA. If the biopsy indicates that GCA is unlikely, the corticosteroid dose can be reduced over a few days to a dose more appropriate for managing PMR alone.
High dose oral prednisone is recommended for patients with suspected GCA
Most guidelines recommend oral prednisone 40 – 60 mg (usually 60 mg in practice), once daily (or 1 mg/kg for patients with a BMI ≤ 18.5 kg/m2).12, 13 For patients with vision loss at presentation, discuss with a rheumatologist or ophthalmologist the suitability of a course of intravenous corticosteroids, e.g. methylprednisolone, 1 g/day for three days, followed by oral prednisone.12, 15
The initial dose of prednisone should be maintained until there is adequate clinical response to treatment (in practice this is usually around one week), e.g. reduction in symptoms, before attempting to taper the dose.16 Tapering schedules should be individualised depending on the patient’s symptoms.16 Example regimens:15
- Reduce dose by 10 mg every two weeks until 20 mg/day, then by 2.5 mg every two to four weeks until 10 mg/day, then by 1 mg every one to two months (provided there are no relapses)
- Reduce dose to 15 – 20 mg/day within two to three months, then after one year to ≤ 5 mg/day
N.B. Other treatments may be added to the corticosteroid tapering regimen, e.g. weekly tocilizumab (not funded) or methotrexate (funded; unapproved indication), to improve the likelihood of remission and/or reduce the number of relapses;15, 16 these decisions are usually guided by a rheumatologist or ophthalmologist (if vision loss is involved).
Aspirin may be considered for some patients
Low dose aspirin (e.g. 100 mg/day) may be considered for some patients, e.g. those with a high atherosclerotic risk to reduce the risk of cranial ischaemic complications, unless contraindicated.1, 17 This is not firmly established across all international guidelines and should only be considered on a case-by-case basis usually in conjunction with a rheumatologist. A proton-pump inhibitor may also be required if aspirin is prescribed due to the risk of gastrointestinal bleeding.1
The main goals of treatment for a patient with PMR are to relieve muscle pain and stiffness, to resolve any other significant symptoms and to prevent relapses.2, 4, 11 PMR tends to worsen if left untreated and patients will get progressively stiffer until they are unable to engage in daily activities. With treatment, activity can be maintained and quality of life can be improved.4 Oral corticosteroids are the first-line treatment for patients with PMR and these are often required long-term, e.g. for several months or years.4, 18 Alternative treatments to corticosteroids may be considered for some patients in consultation with a rheumatologist, e.g. if corticosteroids are contraindicated or ineffective (see: “Alternative treatments to corticosteroids”).
Corticosteroids are the first-line treatment
A trial of an oral corticosteroid can be initiated for patients with suspected PMR once other diagnoses have been excluded and laboratory results (e.g. CRP and/or ESR) are available; prednisone is generally recommended.13, 14 International guidelines advise initiating patients on the lowest effective dose of prednisone between the range of 12.5 – 25 mg:13, 18
- 15 mg prednisone is an appropriate starting dose for most patients
- Patients with mild symptoms, relevant co-morbidities, risk factors for corticosteroid-related adverse effects or frailty may require a lower dose
- Patients with severe initial symptoms or a larger body weight may require a slightly higher dose
The dose is usually taken once daily in the morning, however, if a patient experiences significant symptoms by the end of the day, a twice daily divided dose can be trialled (although taking corticosteroids in the evening can disrupt sleep for some people).1, 13
Patients generally respond rapidly to 15 – 20 mg of prednisone per day and can begin tapering the dose after symptoms have resolved, usually after two to four weeks (see: “Treatment duration and tapering of corticosteroids”).1, 6 An inadequate response to prednisone within seven to ten days is uncommon and if this occurs, the diagnosis should be reconsidered. If clinical suspicion for PMR remains, consider trialling a higher dose of prednisone after discussing with the local rheumatology service (see: “Assess the patient’s response to corticosteroid treatment”).1, 13
N.B. Intramuscular corticosteroids are recommended in some international guidelines as an alternative to oral corticosteroids for some patients with PMR (e.g. if they are unable to swallow tablets or have difficulties with adherence to an oral regimen).5, 6, 18 However, the use of intramuscular corticosteroids for PMR is not widely established in New Zealand. See: “Alternative treatments to corticosteroids” for information on other available treatment options for PMR.
Patients should be regularly reviewed to assess their response to treatment and make any changes to their dosing regimen as required, and to monitor for any symptoms and signs of relapse, corticosteroid-related adverse effects or the development of GCA.4, 12 Laboratory tests such as CRP (or ESR), FBC, serum creatinine and electrolytes, HbA1c (if risk factors for diabetes), may be requested as indicated to provide supporting information. However, in practice not all tests will be necessary in each follow-up appointment, and this is based on clinical judgement.
The frequency of follow-up depends on the patient’s corticosteroid tapering regimen and co-morbidities. In the short term, follow-up typically occurs more frequently, e.g. after one to two weeks of initiating treatment or earlier if there has been no response within a few days or if symptoms worsen (see: “Assess the patient’s response to corticosteroid treatment”), and then ideally at weeks three and six, or when there is symptom relapse.13 If the patient is responding well to treatment at these appointments, the frequency of follow-up can be reduced over time, e.g. every three months and then every 6 – 12 months after the first year for the duration of corticosteroid treatment (but become more frequent again if there is symptom relapse).13
Assess the patient’s response to corticosteroid treatment
 Listen to commentary featuring Simon Stebbings about assessing the effectiveness of corticosteroid treatment
Listen to commentary featuring Simon Stebbings about assessing the effectiveness of corticosteroid treatment
Monitoring a patient’s clinical response to prednisone is the most important metric to assess treatment success. This might be a self-reported improvement in pain and stiffness, or an improved ability to perform a task that had been impaired, e.g. the patient is now able to lift their arms above their head to reach a shelf without shoulder pain. Patients generally experience a rapid improvement in symptoms within a few days to one week of treatment initiation which supports a diagnosis of PMR.1, 5, 8 A small study has shown that after four weeks of treatment, approximately half of patients have complete response to 15 mg of prednisone and approximately one-third have a partial response.19 Treatment response may take longer if the initial dose is lower than 15 mg.
Monitoring CRP (or ESR) is often helpful when assessing a patient’s response to prednisone; levels generally decrease within a few days of treatment initiation and normalise within two to four weeks.8, 13 However, levels of inflammatory markers may not always correlate with the patient's symptomatic response. In addition, some patients may have elevated baseline levels; especially patients with obesity, where levels of inflammatory markers may never fall within the normal range which can make their use for monitoring difficult. A pre-diagnosis baseline CRP (or ESR) measurement is useful to compare in this situation if available.
Gradual tapering of the corticosteroid dose can be attempted once there is adequate improvement in the patient's symptoms (see: “Treatment duration and tapering of corticosteroids”).2, 12 If there is not adequate improvement with an appropriate dose of prednisone within one to two weeks, reconsider the diagnosis. If clinical suspicion for PMR remains, consider trialling a higher prednisone dose (usually up to 30 mg) after discussing with the local rheumatology service.1, 6, 13
Treatment duration and tapering of corticosteroids
Once there is adequate response to treatment (i.e. the patient is experiencing fewer symptoms), the corticosteroid dose can usually be slowly and progressively tapered over time.2, 12 A rapid tapering regimen is associated with a high relapse rate and should be avoided (see: “Relapse of PMR symptoms”).4, 8
Dose tapering schedules should be individualised (see below for examples); there is a lack of consensus on the ideal regimen.3, 13 Some guidelines recommend that the tapering regimen is based on a combination of the patient’s symptoms, the risk/presence of adverse effects from treatment and laboratory markers.1, 18 N.B. Corticosteroid dose should not be increased based on a single raised CRP (or ESR) level if the patient is asymptomatic because inflammatory markers may not always correlate with the patient’s symptomatic response.8, 15
Examples of corticosteroid tapering regimens for PMR:
- Once the dose that controls the patient's symptoms, e.g. 15 mg, has been maintained for 2 – 4 weeks:3, 8, 13
- Reduce dose to 12.5 mg/day for 2 – 4 weeks, then;
- Reduce dose to 10 mg/day for 4 – 6 weeks, then;
- Reduce dose by 1 mg every 1 – 2 months or 2.5 mg every 3 – 4 months until discontinuation (depending on symptoms)
- Taper dose to 10 mg/day within 4 – 8 weeks.4, 18 Once remission is achieved, reduce dose by 1 mg/month until discontinuation.4, 18 Alternate day tapering may also be used.4, 18
If tapering is difficult or if the dose cannot be decreased to ≤ 10 mg/day, discuss with or refer the patient to a rheumatologist.6
The optimal treatment duration with corticosteroids for a patient with PMR is unclear in the literature, but it is largely patient-specific and determined by the presence of symptoms.13 While the aim is to stop prednisone use as soon as possible, the average duration of prednisone use is one to two years.1, 13 Some people, however, may require ongoing low dose prednisone, e.g. ≤ 5 mg, to remain symptom free.1, 3
What to do about patients who struggle to make the final corticosteroid dose reduction and continue to require treatment, e.g. > 5 years?
- Consider adding weekly methotrexate to their regimen in consultation with a rheumatologist.1, 2 Once tolerance is established, trial stopping the corticosteroid. If the patient remains symptom-free, attempt to taper off methotrexate (usually over three months).2
- Consider whether any co-morbidities have since developed, e.g. rheumatoid arthritis, non-inflammatory musculoskeletal problems, malignancy, which may require a different approach to treatment.
- Re-consider the diagnosis of PMR if treatment cannot be completely stopped; refer the patient to a rheumatologist if uncertain.6, 18
Relapse of PMR symptoms
 Listen to commentary featuring Simon Stebbings about relapse of PMR symptoms
Listen to commentary featuring Simon Stebbings about relapse of PMR symptoms
Relapse occurs in approximately half of all patients with PMR and is most common within two years of diagnosis.1, 2, 4 Female sex, elevated inflammatory markers, high platelet count and peripheral arthritis at diagnosis have been associated with a higher risk of relapse.2, 4, 15 Relapse of PMR symptoms is more likely to occur when the corticosteroid dose is < 5 mg per day, when the dose is rapidly tapered and when the corticosteroid has been discontinued.4, 8 Higher initial corticosteroid doses have been reported as a predictor of future relapse.13
Clinical symptoms and signs are the primary markers for relapse with laboratory markers providing supporting information only.4, 8 If a relapse occurs, re-evaluate for alternative diagnoses,13 and:
- If the patient’s symptoms recur during the tapering period, increase the dose to the last effective dose (pre-relapse dose) for some time* before re-attempting a gradual taper.1, 8, 18
- The pre-relapse dose is generally sufficient to manage the patient’s symptoms, however, further increases in dose may be required for some, e.g. an additional 2.5 – 5 mg8
- If the patient has been completely tapered off the corticosteroid when the relapse occurs, re-initiate treatment at the lowest effective dose and then taper as tolerated13
*Some sources report that the dose taper should resume within four to eight weeks, while others recommend to wait at least three to six months or longer1, 18
Adding methotrexate to the treatment regimen
After two relapses, an alternative treatment (usually oral methotrexate, 10 – 20 mg in practice, once weekly; unapproved indication) may be considered in consultation with a rheumatologist.5, 8, 13 There is some evidence that people taking both corticosteroids and methotrexate have a lower risk of relapse compared to those taking corticosteroids alone.5 Regular laboratory monitoring of FBC, creatinine and LFTs is required due to the potential adverse effects of methotrexate.2, 20 To reduce the risk of adverse effects, methotrexate should be prescribed with folic acid.2 Methotrexate is usually continued until the corticosteroid can be tapered without the patient experiencing symptoms.2 Once the corticosteroid has been discontinued, methotrexate can usually be tapered over three months.2
Alternative treatments to corticosteroids
 Listen to commentary featuring Simon Stebbings, about alternative treatments to corticosteroids for patients with PMR
Listen to commentary featuring Simon Stebbings, about alternative treatments to corticosteroids for patients with PMR
In rare cases, some patients with mild PMR may be able to manage symptoms with NSAIDs alone, however, this should not be considered as a standard treatment choice. Short-term NSAID use can be useful alongside prednisone in some patients with pain related to co-morbidities, e.g. osteoarthritis.2, 18
Treatment decisions for a patient with PMR who requires an alternative to corticosteroids are usually guided by a rheumatologist.3, 6
Disease modifying antirheumatic drugs (DMARDs), e.g. methotrexate, leflunomide, may be added to the corticosteroid regimen for some patients (e.g. those at high risk of relapse or after two relapses), or they can be used as an alternative if corticosteroids were ineffective, contraindicated or if significant adverse effects were experienced.6, 12, 18 Methotrexate (oral usually 10 – 20 mg in practice, once weekly; unapproved indication) is typically the DMARD of choice, although data to support its use are limited.12, 18 Methotrexate is most effective when added to the corticosteroid regimen early in the disease course.6, 12 N.B. Folic acid should always be prescribed with methotrexate to reduce the risk of adverse effects.2
Emerging treatments. There is evidence that biologic medicines, e.g. tocilizumab and sarilumab which inhibit interleukin-6 (a major driver of the CRP response), are an effective treatment for PMR allowing earlier reduction in dose and cessation of corticosteroids.5, 21 Tocilizumab is available in New Zealand, although is not indicated or funded for PMR (it is funded with Special Authority for some other conditions, e.g. rheumatoid arthritis). Sarilumab was approved by the United States Food and Drug Administration in March, 2023, for the treatment of PMR, however, it is not currently approved or available in New Zealand.
With prompt treatment, prognosis is generally good for most patients with PMR.2, 5 Pain and functional limitations, e.g. stiffness, often severely impact quality of life but these are generally controlled or resolve with treatment. There is no evidence that PMR causes premature mortality.4, 12 Morbidity associated with PMR typically occurs from adverse effects of long-term corticosteroid use (see below).4, 12 Long-term treatment decisions should consider the adverse effects associated with prolonged corticosteroid use on a case-by-case basis.
Long-term adverse effects of corticosteroids
It is common for people taking corticosteroids to experience adverse effects which tend to be dose and duration dependent; up to 81% of people taking corticosteroids for PMR experience adverse effects within the first year of treatment.3, 8 Monitoring for, preventing and treating any adverse effects of corticosteroids (Table 1) is therefore an important part of the long-term follow-up of patients with PMR.2, 3
Table 1. Common corticosteroid-related adverse effects.
| Corticosteroid-related adverse effect |
Description |
Action |
Reduced bone mineral density |
Long-term corticosteroid use reduces bone mineral density and increases the risk of fracture and osteoporosis.3, 15 |
Consider bone protection for patients taking corticosteroids.5, 8 Generally this will include vitamin D supplements with advice for the patient to ensure adequate intake of dietary calcium.13, 15 If dietary calcium is inadequate, consider supplementation.13 Bone strengthening exercises may also be recommended.15
Bisphosphonates* may be prescribed for patients at high risk of fractures.8, 13 Also consider whether referral for a bone mineral density scan (DEXA) is indicated.3, 15 Check local HealthPathways for specific advice.
*From 1 March, 2023, Special Authority funding restrictions for zoledronic acid have been removed, i.e. there is no longer a requirement for bone mineral density scanning
For further information on bisphosphonates, see: https://bpac.org.nz/2019/bisphosphonates.aspx |
Changes in weight |
People taking corticosteroids can experience an increase in BMI and an altered fat distribution (cushingoid appearance).3, 15 |
Monitor weight/BMI and encourage lifestyle modifications, e.g. healthy diet and physical activity.3, 15
If a patient experiences significant weight changes or cushingoid features, consider discussion with a rheumatologist about adding an alternative treatment to the current regimen to reduce the daily corticosteroid dose.15 |
Diabetes |
Corticosteroid use has been associated with glucose intolerance, elevated fasting glucose levels and an increased risk of type 2 diabetes.3, 15
It can be difficult to manage a patient with PMR and diabetes as corticosteroids can affect insulin dosing requirements and patients tend to have poorer glycaemic control. |
Monitor HbA1c levels if there are risk factors for diabetes, and encourage lifestyle modifications, e.g. healthy diet and physical activity.3, 15 Optimise pharmacological treatments if required.3, 15
If a patient develops new-onset diabetes, reconsider whether their PMR is active by trialling a lower corticosteroid dose.15 Stopping the corticosteroid might resolve the induced diabetes for some patients. If the patient’s diabetes is uncontrolled and the PMR is active, initiate treatment for diabetes. An alternative to oral prednisone, e.g. local corticosteroid injections, methotrexate, may be needed for some patients with diabetes and active PMR after consultation with a rheumatologist. |
Gastrointestinal |
An increased risk of gastrointestinal-related adverse effects such as dyspepsia or peptic ulcer disease have been associated with long-term corticosteroid use.3, 13 |
Consider prescribing a proton pump inhibitor (e.g. omeprazole) to prevent steroid-induced gastritis particularly for patients at high risk of peptic ulcers, e.g. those who take aspirin.3, 12
Ensure patients are taking their corticosteroid with (or just after) food to reduce gastrointestinal effects. |
Adrenal insufficiency |
Long-term corticosteroid use can inhibit function of the hypothalamic-pituitary-adrenal axis, leading to adrenal insufficiency.3, 22 |
Ensure slow tapering when the dose of prednisone is low;22 some patients may be able to self-titrate.
Educate patients on the possible symptoms of adrenal insufficiency and advise them about what to do if this occurs, e.g. increase dose of prednisone.22
Consider increasing the corticosteroid dose during times of physical stress, e.g. acute illness, trauma or surgery.3, 22
Adrenal function testing is not usually required in primary care, but may be used in some cases, e.g. if rapid withdrawal is required or if there is difficulty discontinuing use.3 |
Visual disturbances |
Corticosteroid use is associated with an increased risk of visual disturbances due to conditions such as cataracts and glaucoma.3, 15 |
Educate patients on the possible adverse effects to their vision, and advise them to seek a medical or optometrist assessment if they experience worsening vision or other visual disturbances.3 |
Cardiovascular |
There is a possible increased risk of cardiovascular-related adverse effects, e.g. dyslipidaemia, hypertension, atherosclerosis, myocardial infarction.3, 15 |
Screen for cardiovascular risk factors, measure fasting lipid levels and blood pressure and encourage lifestyle modification, e.g. healthy diet and physical activity.3, 15 Initiate pharmacological treatments as required.3 |
Infection |
There is a possible increased risk of infection due to the immune suppressive effects of systemic corticosteroids.3, 4 |
Recommend that patients are up to date with vaccinations, e.g. influenza, COVID-19, and educate patients about their increased risk of infection and discuss infection prevention measures.3, 4 Patients taking high dose corticosteroids must avoid live vaccines.16 |
Skin changes |
Long-term corticosteroid use has been associated with changes to the skin, including an increased risk of skin atrophy, acne, alopecia, susceptibility to bruising and hirsutism.3, 4 |
Educate patients on potential changes to their skin and give general skin health advice.3 If changes occur, lower the dose of corticosteroid if possible. |
Mood disturbances |
Rarely, an increased risk of insomnia, violence, depression, psychosis and other neuropsychiatric disorders has been associated with long-term corticosteroid use (mood disturbance can also occur with short courses of corticosteroids if the dose is high).3, 4 |
Educate patients on the possible risk of mood disturbance, screen for relevant disorders (as indicated) and initiate non-pharmacological and pharmacological treatments (if required).3
If a patient develops insomnia, check that the dose of the corticosteroid is being taken in the morning.3 |





