Published: May, 2025 | Review date: May, 2028
This audit focuses on optimising allopurinol treatment in patients with gout, to ensure they are being treated to target.
Gout is the most common form of inflammatory arthritis. It is associated with monosodium urate crystals accumulating in joint fluid, cartilage, bones, tendons and other tissues. The inflammatory response to these crystals results in gout flares, characterised by painful, red, hot, swollen joints, usually in the first metatarsophalangeal joint. Over time, the duration and frequency of these flares may increase, resulting in chronic gouty arthritis and subcutaneous deposits of crystals, referred to as tophi, which can lead to joint destruction and musculoskeletal disability.
The goal of urate-lowering treatment is to reduce serum urate levels below the saturation point to dissolve urate crystals, thereby preventing future gout flares. The recommended serum urate levels are:
- < 0.36 mmol/L – for most patients; or
- < 0.30 mmol/L – for patients with severe gout, e.g. those with tophi, chronic gouty arthritis or frequent flares
Allopurinol is the first-line urate-lowering medicine and should be discussed with all patients with gout once a diagnosis has been made (even if it is not prescribed straight away). The dose of allopurinol must be slowly up-titrated to achieve the treatment target, and serum urate testing should be requested prior to each dose adjustment. Once the patient has reached the treatment target, serum urate levels should be monitored every 6 – 12 months.
For further information on the diagnosis and management of gout, see: www.bpac.org.nz/2025/gout.aspx
Summary
This audit identifies patients with gout who are prescribed allopurinol and determines if they have been treated to target.
- Allopurinol doses should be titrated to achieve a target serum urate level < 0.36 mmol/L
- Serum urate levels should be tested every 6 – 12 months, once the serum urate target has been reached, as part of routine patient monitoring
Recommended audit standards
Ideally, all patients with gout should be treated to a target serum urate level, but in practice this is not always achievable, e.g. if flares are controlled and urate levels are only slightly above target despite the maximum tolerated dose, some patients may choose to persist with allopurinol alone, rather than taking an additional medicine to further lower levels.
Ideally, 100% of patients with gout should have had a serum urate level test within the past 12 months. A reasonable audit standard is for 80% of the patient sample to be treated to target, i.e. serum urate level < 0.36 mmol/L. This target may be lower in the first cycle of the audit, with an increase in the number of patients who have reached the target urate level after the second cycle.
 Alternatively, consider a “working audit” where the data sheet is completed opportunistically during consultations for any reason (including repeat prescriptions) with an eligible patient until the required number of patients has been reached. If conducting a working audit and there is no record of a serum urate level < 0.36 mmol/L within the previous 12 months, a review should be undertaken at the time (including requesting serum urate testing), or a future appointment booked.
Alternatively, consider a “working audit” where the data sheet is completed opportunistically during consultations for any reason (including repeat prescriptions) with an eligible patient until the required number of patients has been reached. If conducting a working audit and there is no record of a serum urate level < 0.36 mmol/L within the previous 12 months, a review should be undertaken at the time (including requesting serum urate testing), or a future appointment booked.
Eligible patients
All patients diagnosed with gout who have been prescribed allopurinol for ≥ 12 months are eligible for this audit.
Identifying patients
You will need to have a system in place that allows you to identify eligible patients and audit their clinical notes. Many practices will be able to do this by running a ”query” through their PMS to find all patients prescribed allopurinol. Exclude patients taking allopurinol for other reasons.
If conducting a working audit, fill in the data sheet when you have a consultation for any reason with an eligible patient until the required number of patients has been reached.
Sample size
The number of eligible patients will vary according to your practice demographic. If a large number of results are returned, a sample size of 30 patients is sufficient for this audit. A smaller sample size may be necessary if conducting a working audit to complete it within your planned time frame.
Criteria for a positive outcome
For a positive result, the patient’s clinical notes should contain a record of a serum urate level within the previous 12 months, and this level should be < 0.36 mmol/L. If the patient does not have this in their clinical notes, they should be flagged for review to consider how treatment can be optimised.
 If conducting a working audit and there is no record of a serum urate level < 0.36 mmol/L within the previous 12 months, a review should be undertaken at the time (including requesting serum urate testing), or a future appointment booked.
If conducting a working audit and there is no record of a serum urate level < 0.36 mmol/L within the previous 12 months, a review should be undertaken at the time (including requesting serum urate testing), or a future appointment booked.
Data analysis
Use the sheet provided to record your data. The percentage of patients being treated to target can be calculated by dividing the number of patients who had a serum urate level < 0.36 mmol/L in the past 12 months and dividing it by the total number of patients audited.
Optional: record the patient’s serum urate level to assess how far from target they are.
Clinical audits can be an important tool to identify where gaps exist between expected and actual performance. Once completed, they can provide ideas on how to change practice and improve patient outcomes. General practitioners are encouraged to discuss the suitability and relevance of their proposed audit with their practice or peer group prior to commencement to ensure the relevance of the audit. Outcomes of the audit should also be discussed with the practice or peer group; this may be recorded as a learning activity reflection if suitable.
The Plan, Do, Study, Act (PDSA) model is recommended by the Royal New Zealand College of General Practitioners (RNZCGP) as a framework for assessing whether a clinical audit is relevant to your practice. This model has been widely used in healthcare settings since 2000. It consists of two parts, the framework and the PDSA cycle itself, as shown in Figure 1.
1. The framework
This consists of three questions that help define the “what” and “how” of an improvement project (in this case an audit).
The questions are:
- "What are we trying to accomplish?" – the aim
- "How will we know that a change is an improvement?" – what measures of success will be used?
- "What changes can we make that will result in improvement?" – the concept to be tested
2. The PDSA cycle
This is often referred to as the “engine” for creating, testing and carrying out the proposed changes. More than one cycle is usually required; each one is intended to be short, rapid and frequent, with the results used to inform and refine the next. This allows an ongoing process of continuous learning and improvement.
Each PDSA cycle includes four stages:
- Plan – decide what the change to be tested is and how this will be done
- Do – carry out the plan and collect the data
- Study – analyse the data, assess the impact of the change and reflect on what was learned
- Act – plan the next cycle or implement the changes from your plan
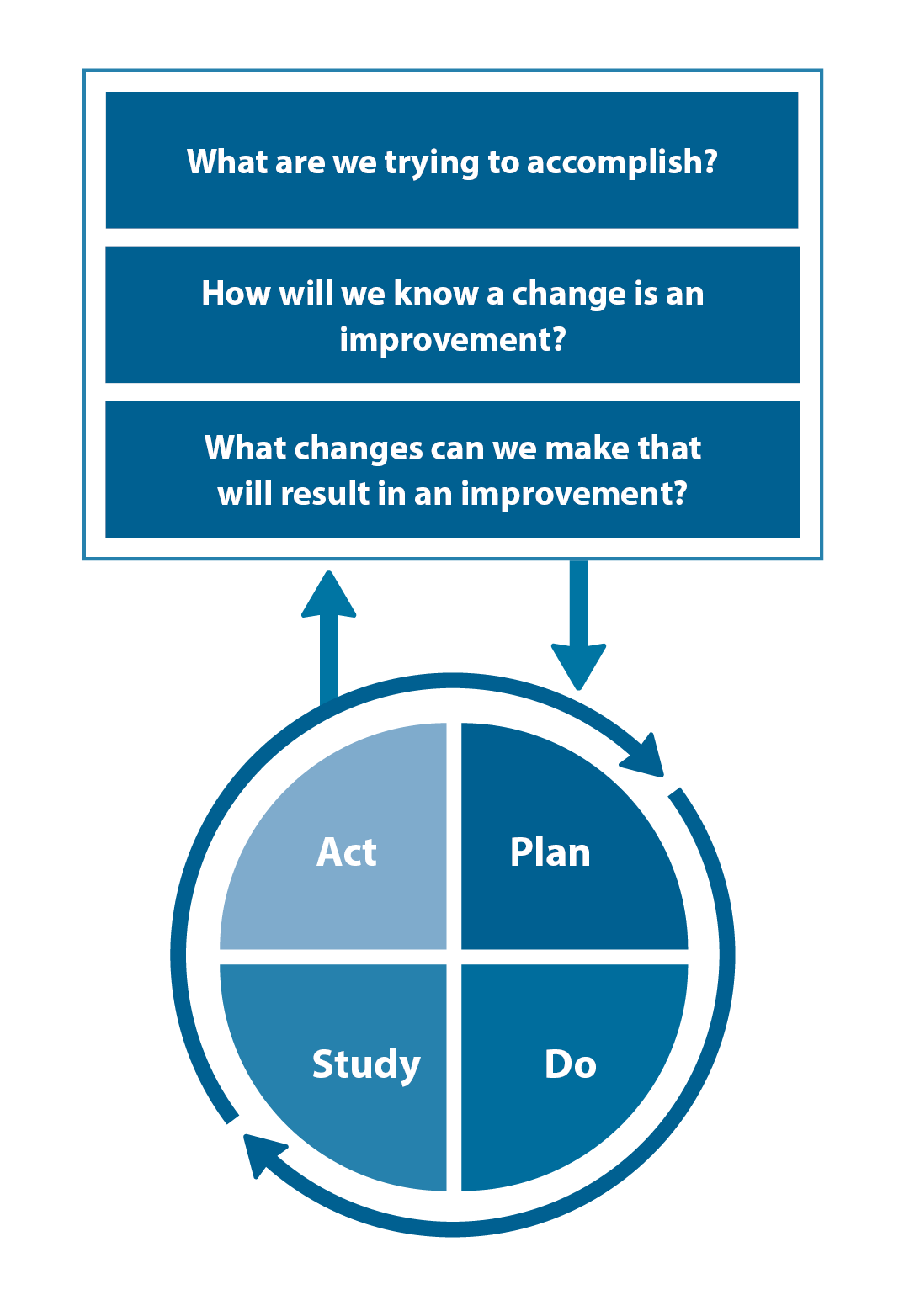
Figure 1. The PDSA model for improvement.
Source: Plan, Do, Study, Act (PDSA) cycles and the model for improvement

Claiming credits for Te Whanake CPD programme requirements
Practice or clinical audits are useful tools for improving clinical practice and credits can be claimed towards the Patient Outcomes (Improving Patient Care and Health Outcomes) learning category of the Te Whanake CPD programme, on a two credit per learning hour basis. A minimum of 12 credits is required in the Patient Outcomes category over a triennium (three years).
Any data driven activity that assesses the outcomes and quality of general practice work can be used to gain credits in the Patient Outcomes learning category. Under the refreshed Te Whanake CPD programme, audits are not compulsory and the RNZCGP also no longer requires that clinical audits are approved prior to use. The college recommends the PDSA format for developing and checking the relevance of a clinical audit.
To claim points go to the RNZCGP website: www.rnzcgp.org.nz
If a clinical audit is completed as part of Te Whanake requirements, the RNZCGP continues to encourage that evidence of participation in the audit be attached to your recorded activity. Evidence can include:
- A summary of the data collected
- An Audit of Medical Practice (CQI) Activity summary sheet (Appendix 1 in this audit or available on the
RNZCGP website).
N.B. Audits can also be completed by other health professionals working in primary care (particularly prescribers), if relevant. Check with your accrediting authority as to documentation requirements.





