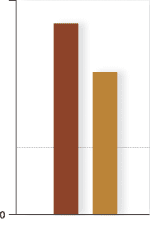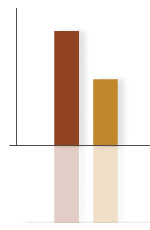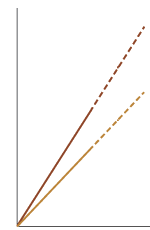| Essentials | 
|
Full colour PDF of the pages as they appeared in ‘best practice’.
 Printer friendly PDF.
Printer friendly PDF.
Critical appraisal of
Drug advertising
Pharmaceutical companies spend large amounts of money on advertising and promotional activities to persuade doctors to prescribe their products. In this article we provide a quick guide to some of the issues to consider when reading drug advertising material or talking to pharmaceutical representatives.
| How cleverly by divers means By graphical skills and careful schemes Those who our medicines prepare. Would thus the innocent ensnare, The latest tablet we are told, Will stop our organs growing old, And, furthermore by slow release, Will thus its healing powers increase. How well advised are those who opt, A cautious response to adopt. Hospital, Polly Toynbee, Hutchinson London 1977 |
The standards of drug advertising in New Zealand are self-regulated by a code of practice issued by the Researched Medicines Industry.
The Code of Practice is available from:
http://www.rmianz.co.nz/pdfs/COP2006/Principle6.pdf
Supporting Evidence
Check that claims are supported by high quality randomised controlled trials or meta-analyses rather than surrogate end-points or ‘class effects’. Many references and supporting claims are of poor quality such as reports from meetings, symposia, journal supplements or very small studies. Journals cited can sometimes be very obscure indicating the possibility that the study has not met high quality peer review standards. References should be accessible and produced on request and this is often a problem when the source is ‘data on file’ i.e. information held by the company which is not in the public domain.
Words and phrases
Single words or phrases are often used to empower prescribers without any underpinning science or evidence. The word ‘new’ is often used, but as in the case of the COX-2 inhibitors ‘new’ is not always better.
Commonly misused shortcuts for choosing therapies1
|
Other frequently used words and phrases include;
- Power
For example, ‘power in your hands’, ‘power to prescribe’. Often employed in various contexts such as giving you (the GP) the power to do something for your patient. The implication is that if you don’t prescribe the drug you might deny the patient a useful treatment. - Potency
Just because a drug is more potent (less required for clinical effect) doesn’t mean it is more effective or safer. Readers are often falsely drawn in to linking ‘higher potency’ with improved clinical effect. - New, Improved, Better, New Formulation
Newer is not always better and we actually have very limited knowledge about the safety of new drugs. Statements such as ‘better’ or ‘improved’ need qualification with high level evidence.
Red Herring effect
Statements can be used that have no link or association with the clinical effectiveness of the drug. These are used to sell a unique property of the drug but this may have no relevance to the therapeutic effect. Some examples;
This drug:
- Has a unique carboxyl group on the terminal chain
- Is the most powerful drug in its class
- Penetrates the bacterial cell wall better
- Binds specifically to tissue enzymes
A particularly common red herring is a statement that the drug is the ‘most widely’ prescribed in a particular country or the number of prescriptions dispensed worldwide. Although this may be reassuring clinical effectiveness still needs to be backed up with good evidence.
All such statements should be related to actual patient outcomes otherwise they are meaningless.
Appeal to Authority
Often eminent physicians from reputable institutions are quoted as having used the drug in their practice or they endorse its use.
‘Dr. Perfect from Stargaze University uses this drug’
Increasingly media personnel are being used to endorse and market pharmaceuticals.
Colours, pictures and emotion
Often 70–80 % of an advert is a picture or graphic. A significant amount of design work is involved to market the message and catch the eye. Emotion and colour are large components in marketing e.g. the white coat, the caring physician, the concerned patient and happy families.
Graphs and statisticsGraphs and charts are favourite tools of the drug advertiser. These often appear in glossy brochures and presentations and can be manipulated to over emphasise or dramatise effects. Some things to check are whether
Benefits are nearly always presented as Relative Risk or Relative Risk Reduction as prescribers are more likely to be impressed by these figures than Absolute Risk Reduction or NNTs. Ask representatives about the absolute benefits of treatment. It should be possible to calculate these figures yourself (see BPJ Issue 2) from the figures presented or the references. p-values should have associated 95% confidence intervals so that the strength of the relationship can be assessed rather than just a statistically significant difference. In summary awareness and use of the above prompts allows a quick evaluation of drug advertising so you can cut through the marketing hype to assess the real value of the therapy. References and bibliography
|
|